Substrate pH

pH determines micronutrient solubility in the root substrate:
• low pH = very soluble
• high pH = less soluble
►
Plant takes up soluble nutrients through roots
► Nutrients transported into leaves and growing points:
• Excess = toxicity
• Adequate = healthy
• Insufficient = deficiency
► Toxicity or Deficiency results in stunted growth, poor plant appearance, and un-saleable plants
________________________________________
. Why is pH important?
The most common nutritional problems occur in greenhouse container-grown crops when pH of the root substrate (also called potting mix or growing substrate) is outside the acceptable range. Substrate- pH measures acidity (low pH = acid) or basicity (high pH = basic, also called alkaline) of a root substrate, which affects a chain of events that ultimately affects plant health.
Plants take up dissolved nutrients through their roots. Substrate-pH drives chemical reactions that determine whether nutrients are either available for root uptake (i.e. soluble and dissolved) or unavailable for uptake (i.e. insoluble and solid). The most important nutrient affected by substrate- pH is iron (Figure 1). Iron, phosphorus and the micronutrients manganese, copper, zinc, and boron also decrease in solubility at high pH. Molybdenum increases in solubility at high pH.
The acceptable range for most crops growing in a soilless substrate is 5.6 to 6.4, because in this range micronutrients are soluble enough to satisfy plant needs without becoming toxic.
Recognizing the problem
Iron is required by plants to produce chlorophyll (the pigment in leaves which gives plants their green color). At high substrate-pH, iron (as well as most other micronutrients) becomes less soluble in the root substrate, resulting in lower uptake by the plant
(Figure 1). Because iron is not mobile within the plant, deficiencies induced by high pH tend to show chlorosis (yellowing caused by lack of chlorophyll) in the growing
tip. Sometimes, the chlorosis is over the entire leaf, but it can also be interveinal (leaf veins are green, but remaining tissue is yellow) as shown with the picture above of petunias grown at high pH. As the deficiency becomes more severe, plants lose vigor and the color of the chlorosis changes from yellow to almost completely white with necrotic (dead) areas forming at the growing points.
At low substrate-pH,
iron and manganese are highly soluble in the root substrate. Excess micronutrients can accumulate in plant tissue and cause chlorosis and necrosis (dead tissue) on leaf margins and as leaf spots, as shown in these geranium and marigold leaves. The damage tends to occur in older leaves because the longer a leaf grows on the plant, the more time it has to accumulate excess micronutrients.
Why do Substrate-pH problems arise?
Reasons that substrate-pH can be too high or low include:
3A. Poor buffering of soilless substrate. The move away from use of soil in greenhouse container substrate has many benefits (uniformity, consistency, aeration, sterility) but results in less buffering (chemical resistance to pH change) than soil.
A pH unit change within one week can sometimes occur with soilless substrate. Substrate-pH can drift up or down depending on the balance of factors including water alkalinity, lime activity, acidification of the substrate by plant roots, and use of an acid or basic reaction fertilizer. As a result, it is important not just to blame problems on the substrate or
fertilizer, but rather to also understand how grower management can cause pH to change over time.
3B. Limestone. Limestone is mixed into substrate to raise pH to around 6.0 because both peat and bark are acidic. Limestone sources differ in their composition, particle size, and hardness, which causes them to vary in their reactivity (i.e. how many pounds per cubic yard are required to raise pH at the start of the crop), and also in how long they continue to react during crop growth.
If the incorrect type or quantity of lime is used during mixing of the substrate, pH can either be out of range at the start of the crop, or it can drift over time. If mixing your own substrate, (a) consult a fertilizer or substrate company to obtain a suitable type of lime, (b) run small batch tests to check how much lime is needed to bring pH up to the target level, and (c) if you change the source of lime, peat, bark, or vermiculite you will need to re-test your recipe.
If you consistently run into problems with high or low substrate-pH, and you have correctly matched the fertilizer type with water alkalinity, consider changing the lime type or rate.
3C. Wide range in crops. Species differ in their nutritional needs, and can be separated into three nutritional groups based on their efficiency at taking up iron from the root substrate.
- Petunia group: Also known as iron- inefficient species, prone to iron deficiency at high substrate-pH, especially when combined with low fertilizer concentration. Grow at a lower substrate-
pH range of 5.4-6.2 to maximize iron solubility. This group is often misdiagnosed as a “high feed” or “high iron” group. They do not necessarily require higher
rates of fertilizer or iron, but are especially sensitive to high pH and the need for adequate iron. Examples include bacopa, basil, blueberry, calibrachoa, diascia, nemesia, pansy, petunia, snapdragon, vinca.
- General group: Not prone to pH related problems. Grow at a moderate pH range of 5.8-6.4. Examples include chrysanthemum, impatiens, ivy geranium, poinsettia, tomato.
- Geranium group: Also known as iron-efficient species, prone to iron/ manganese toxicity at low pH, especially when combined with high fertilizer concentration. Grow at a higher substrate-pH range of 6.0-6.6 to limit the
solubility of iron and manganese. Examples include marigold, seed and zonal geranium, New Guinea impatiens, and lisianthus.
Figure 1. The effect of growing petunia at different substrate-pH levels on
(A) soluble iron content in the soil solution, (B) iron content in the leaf tissue, and (C) leaf chlorophyll content. As pH increased there was decreased iron uptake. At the highest pH, substrate and tissue iron levels were the lowest, and the plants showed classic iron deficiency symptoms (chlorosis due to the lack of chlorophyll, see photo above right on this page). Research by Brandon Smith and Paul Fisher, Univ. of New Hampshire, and William Argo, Blackmore Co.


. Fertilizer type.
You cannot measure
the acid or basic reaction of a water-soluble fertilizer by measuring the pH of the stock tank or the solution coming out of the end of the hose. Rather, it is the tendency of a water-soluble fertilizer to change substrate-pH over time after the fertilizer interacts with plants or microbes.
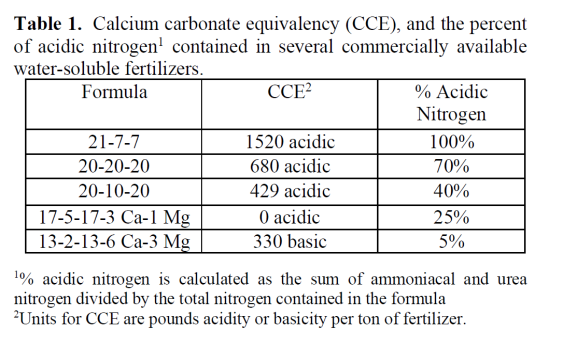
The label on a fertilizer bag usually provides information on the acid or basic reaction of a water- soluble fertilizer expressed as an acidic or basic “calcium carbonate equivalency” (CCE). The CCE is a relative measure of the tendency of the fertilizer to raise or low lower substrate-pH (Table 1).
More importantly, the label tells the type and percentage of the different forms of nitrogen
(ammonium, nitrate, or urea), as well as the percentage of the other nutrients contained in the fertilizer.
In general, ammoniacal and urea nitrogen are acidic, and tend to drive the substrate-pH down, whereas nitrate nitrogen is weakly basic and tends to drive the substrate-pH up.
Several factors are important when using fertilizers to raise or lower substrate-pH:
- Nitrate only increases substrate-pH when the fertilizer is taken up by plant roots. Therefore, if plants are small, or are stressed and not growing, nitrate has little influence on substrate-pH.
- Ammonium can cause the substrate-pH to go down even if the plant is small or is not growing, because soil bacteria acidify the substrate through a process termed nitrification.
- Ammonium is less effective at lowering substrate-pH in cool, saturated soil because nitrification is inhibited. In addition, ammonium toxicity in plants can occur in cool, wet conditions because plants are more likely to take up excess ammonium.
- Sometimes ammonium will not drop substrate-pH at all because other factors (especially excessive lime rates in the substrate or high water alkalinity) can have a stronger effect on pH than the fertilizer, counteracting the pH effects of the fertilizer.
Table 1. Calcium carbonate equivalency (CCE), and the percent of acidic nitrogen1 contained in several commercially available water-soluble fertilizers. 1% acidic nitrogen is calculated as the sum of ammoniacal and urea nitrogen divided by the total nitrogen contained in the formula 2Units for CCE are pounds acidity or basicity per ton of fertilizer.
Irrigation water alkalinity.
Irrigation water pH affects chemical solubility of solutions, but has little effect on substrate-pH.
Instead, substrate-pH is affected by water alkalinity, which is a measure of the basic ions, mainly bicarbonates and carbonates, dissolved in the water. Alkalinity can be thought of as the “liming content” of the water, and irrigating with a high alkalinity water (above 150 ppm CaCO3 of alkalinity) can cause substrate-pH to increase over time.
Options for alkalinity management are:
Alkalinity can be reduced by injecting strong mineral acids (like sulfuric or phosphoric acid) into the irrigation water.
It may be feasible to change or blend water sources. Rain water collected in cisterns or ponds and water purified using reverse- osmosis contain little if any alkalinity.
Matching the alkalinity of the water to the reaction produced by the fertilizer is the most important decision growers can make to maintain a stable pH. For example, a low- alkalinity water should be balanced with a “basic fertilizer” (Table 1, containing low levels of ammoniacal nitrogen). A high- alkalinity water can be balanced with an “acidic fertilizer” (one high in ammoniacal nitrogen). Problems can occur when using ammonium-based fertilizers in cold weather (see 3D above).
1. Substrate-pH and electroconductivity (EC) are the most important soil measurements. This article also mentions pH and EC test options for hydroponics.
If you can maintain substrate-pH and substrate-EC within acceptable limits and select an appropriate growing substrate and fertilizer, you will avoid 95% of nutritional problems.
- Substrate-pH affects how soluble fertilizer nutrients will be in the growing substrate, especially micronutrients such as iron, and soluble nutrients are the only ones available for uptake by roots.
- Substrate-EC measures how much nutrients are in the growing substrate. Substrate-EC is also increased by non-fertilizer ions such as sodium and chloride (table salt) that do not help plant growth.
You could send samples into a lab for pH and EC testing, but unless you have a very small greenhouse it is better to purchase a pH and EC meter and use the laboratory for more detailed analysis of water quality and specific nutrient levels.
The advantages of in-house testing of pH and EC are the low cost and the ability to quickly take a lot of samples on a regular basis.
The goal is to keep the pH and nutritional levels within an acceptable range and to spot problem trends early on. This is a far better strategy than
having to take dramatic steps to rescue stressed crops.
You can therefore use substrate-pH and substrate- EC results to make fertilizer decisions in a simple, systematic way. If pH is too low (acidic), then basic fertilizers are needed. If the pH is too high (basic), then acidic fertilizers can be applied. If the EC is too low (deficient in nutrients) then fertilizer concentration can be increased. If the EC is too high, then apply less fertilizer and perhaps remove fertilizer from the pot by leaching.

In general, there is no one “best” method for measuring substrate-pH or EC in the greenhouse. Consider how much experience you have with a particular method as well as how much help and advice you can get from other people that are close by such as other growers, extension agents, universities, or soil testing laboratories. The chart above, and descriptions of each method may also help you in choosing between methods.
In container substrates:
- Pour-through: Distilled water is poured into the top of the pot displacing a small amount of the soil solution. The solution is then collected at the base of the pot. This method is good for rapid, non- destructive measurement in the greenhouse, and is especially good for large pots or pots that contain slow-release fertilizer prills which can break open and give false readings with the 1:2 or Saturated Paste.
- 1 part soil:2 parts water dilution (“1:2”): Soil is removed from the pot, mixed with twice the volume of water, and pH and EC of this solution are
measured. This method is good for running many samples, and guidelines are well established. In Europe and some other countries, a similar method is used but with more or less water meaning that the EC guidelines differ because of dilution.
- Saturated Paste Extract (“SPE”): A small sample of soil is removed from the pot and enough water is added to the soil sample to saturate air spaces. This is generally the most consistent method but also takes slightly more time to prepare. This method is used by commercial labs, and is also known as the saturated media extract (SME) method.
- Plug squeeze method: An hour after irrigation, or when a plug is still close to saturation from misting, the soil solution is squeezed out of the plug and collected. This method is used for plugs where samples are difficult to handle with the above methods.
- Direct measurement: Sensors with fine tips are placed directly into the substrate. This method is used mainly with plugs or saturated hydroponic substrates such as rockwool or coconut coir, using sensors that can measure directly into the growing substrate. This method is faster but more variable (especially for pH) than other methods.
In hydroponic culture:
- Drip and drain: When growing in enclosed bags or a highly porous substrate such as volcanic rock, test the pH, EC and volumes of both the applied (drip) solution and the leachate (drain) solution and compare trends over time. Many growers have a target leaching fraction (drain volume/drip volume) that is measured and maintained to manage EC level.
pH and EC targets for the drip solution related to the input from the fertilizer injector (typically EC 1 to 2.5 mS/cm depending on crop and pH 5.8 to 6.2). Acceptable pH target for the drain is typically around 6.0 and is managed with acid or base injection of the drip solution. A crop-specific upper EC limit is defined (typically
between 2.5 and 3.5 mS/cm) and is managed using both the drip EC and leaching fraction.
- Reservoir: In liquid hydroponic culture such as nutrient film technique (NFT) or deep water culture there is very little substrate. Therefore, the reservoir itself is sampled, often with continuous inline monitoring.
The pH is adjusted using acid or base injection and a low EC is corrected by adding stock solution. When EC is excessively high, a portion of the nutrient solution is usually discharged and the reservoir is diluted from fresh water. Typical pH target is 5.8 to 6.2 for all crops, and EC 1.5 to 2.5 mS/cm depending on the crop.
Consistency is the key
In order to make decisions, you need good data. This means purchasing a good quality pH and EC meter and calibrating the meter before each use (see our article on this topic). Consistency starts with having a single, trained person performing the test. Choose one method and stick with it. Follow the detailed sampling steps described in our other articles.
Interpreting your results
After you measure the sample, use the correct standards to check whether pH and EC are in the optimum range.
Each method can give different results, especially for EC, because methods vary in how much the soil solution is diluted. For example, diluting a sample by half with distilled water will result in half the EC level. pH is less sensitive to the testing method, but can still vary by ±0.5 pH units between testing methods. This means that you need to use the correct tables to interpret results and be consistent.
For example, less water is added in the SME method than with the 1:2 (i.e. the 1:2 results in a
more dilute solution). EC levels are therefore lower from a 1:2 soil test compared with an SME soil test.
The optimum substrate-pH (Table 1) varies depending on the tendency of a crop species to have problems at low pH (micronutrient toxicity, for example marigold) or at high pH (micronutrient deficiency, for example petunia). The optimum pH is designed to make nutrients soluble enough for healthy uptake without the risk of excess and toxicity.
Substrate-EC is a measure of the total salt concentration in the extracted solution. The EC measurement does not indicate the concentration of any individual plant nutrient. The only way to determine exactly what ions make up the EC is to use a more detailed commercial laboratory analysis. Caution is needed in interpreting an “optimum EC” when deciding whether plants have received sufficient or excess fertilizer. A low substrate-EC, compared with Table 2, does not necessarily indicate a problem. This is because vigorously- growing plants can rapidly (within hours) take up fertilizer nutrients into the tissue, especially in small containers such as plugs.
Observing the overall appearance of the plant, checking the fertilizer level being applied, and using your grower experience are equally important. Also check for trends from one week to the next. If EC is tending to increase over time this indicates excess fertilizer application. If EC is stable, plant uptake and applied fertilizer are probably in balance. If EC is decreasing then the plant is taking up more nutrients than the fertilizer level currently being applied.
Table 1. Interpretation of substrate pH levels for container grown crops. Values are the same for all substrate testing methods.
| Acceptable range | Examples | |
| Iron-inefficient or “Petunia” Group | 5.4 to 6.2 | Arugula, azalea, bacopa, basil, blueberry, calibrachoa, dianthus, nemesia, pansy, petunia, rhododendron, snapdragon, verbena, vinca, and any other crop that is prone to micronutrient deficiency (particularly iron) when grown at high substrate pH. |
| General Group | 5.8 to 6.4 | Chrysanthemum, cucumber, impatiens, ivy geranium, lettuce, osteospermum, pepper, poinsettia, tomato, and any other crop that is not generally affected by either micronutrient deficiencies or toxicities. |
| Iron-efficient or “geranium” group | 6.0 to 6.6 | Lisianthus, marigolds, New Guinea impatiens, seed geraniums, zonal geraniums, and any other crop that is prone to micronutrient toxicity (particularly iron and manganese) when grown at low substrate pH |
Table 2. Interpretation of substrate electrical conductivity (EC) or soluble salt levels.
| 1:2 Method | Saturated Paste Extract method | Pour-through method | Plug squeeze method | |
| Low fertility | 0 to 0.5 | 0 to 1.0 | 0 to 2.4 | 0 to 2.4 |
| Acceptable range | 0.6 to 1.5 | 1.1 to 3.0 | 2.5 to 5.0 | 2.5 to 4.0 |
| High fertility | >1.5 | >3.0 | >5.0 | >4.0 |
Values are reported in milliSiemens per centimeter (mS/cm). The units of measure for EC can be mMho/cm, dS/m, mS/cm, µM/cm or mMho x 10-5/cm. The relationship is 1 mMho/cm=1 dS/m=1 mS/cm=1000 µS/cm=100 mMho x 10-5/cm.
video-alkalinity of irrigation water ( dissolved Lime CaCO3)
mg/L or ppm bicarbonates

