Passive Transport
Diffusion
This movement of molecules or ions from a region of higher concentration to a region of lower concentration is called diffusion. Molecules that are moving from a region of higher concentration to a region of lower concentration are said to be moving along a diffusion gradient, while molecules going in the opposite direction are said to be moving against a diffusion gradient. When the molecules, through their random movement, have become distributed throughout the space available, they are considered to be in a state of equilibrium. The rate of diffusion depends on several factors, including tem- perature and the density of the medium through which it is taking place.
Except within the area immediately surrounding the source, unaided diffusion requires a great deal of time because mole- cules and ions are infinitesimally small. Something that is less than a millionth of a millimeter in diameter is going to take a long time to move just 1 millimeter, even though the amount of movement may be great in proportion to the size of the particle concerned. In gases, there is a great deal of space between the molecules and correspondingly less chance of the molecules bumping into each other and thus being slowed down. Accordingly, gas molecules occupy a space that becomes available to them relatively rapidly, while liquids do so more slowly, and solids are slower yet.
| Substances diffuse according to their concentration gradient; within a system, different substances in the medium will each diffuse at different rates according to their individual gradients.After a substance has diffused completely through a space, removing its concentration gradient, molecules will still move around in the space, but there will be no net movement of the number of molecules from one area to another, a state known as dynamic equilibrium.Several factors affect the rate of diffusion of a solute including the mass of the solute, the temperature of the environment, the solvent density, and the distance traveled. |

Osmosis
Solvents are liquids in which substances dissolve. Despite the fact that the cytoplasm of living cells is bounded by membranes, it is now well known that water (a solvent) moves freely from cell to cell. This has led scientists to believe that plasma, vacuolar, and other membranes have tiny holes or spaces in them, even though such holes or spaces are invisible to the instruments pres- ently available. It also has led to the construction of models of such membranes (see Fig. 3.11). Membranes through which dif- ferent substances diffuse at different rates are described as semipermeable. All plant cell membranes appear to be semiperme- able.
In plant cells, osmosis is essentially the diffusion of water through a semipermeable membrane from a region where the water is more concentrated to a region where it is less concentrated. Osmosis ceases if the concentration of water on both sides of the membrane becomes equal.
A demonstration of osmosis can be made by tying a membrane over the mouth of a thistle tube that has been filled with a solution of 10% sugar in water (i.e., the solution consists of 10% sugar and 90% water). Fluid rises in the narrow part of the tube as osmosis occurs when the thistle tube is immersed in water
Although the previous simple definition of osmosis serves our purposes, plant physiologists prefer to define and discuss osmosis more precisely in terms of potentials. It is possible to prevent osmosis by applying pressure. Just enough pressure to prevent fluid from moving as a result of osmosis is referred to as the osmotic potential of the solution. In other words, os- motic potential is the pressure required to prevent osmosis.
Water enters a cell by osmosis until the osmotic potential is balanced by the resistance to expansion of the cell wall. Wa- ter gained by osmosis may keep a cell firm, or turgid, and the turgor pressure that develops against the walls as a result of water entering the vacuole of the cell is called pressure potential.
The release of turgor pressure can be heard each time you bite into a crisp celery stick or the leaf of a young head of let- tuce. When we soak carrot sticks, celery, or lettuce in pure water to make them crisp, we are merely assisting the plant in bringing about an increase in the turgor of the cells.
The water potential of a plant cell is essentially its osmotic potential and pressure potential combined. If we have two ad- jacent cells of different water potentials, water will move from the cell having the higher water potential to the cell having the lower water potential.
Osmosis is the primary means by which water enters plants from their surrounding environment. In land plants, water from the soil enters the cell walls and intercellular spaces of the epidermis and the root hairs and travels along the walls until it reaches the endodermis. Here it crosses the differentially permeable membranes and cytoplasm of the endodermal cells on its way to the xylem. Water flows from the xylem to the leaves, evaporates within the leaf air spaces, and diffuses out (tran- spires) through the stomata into the atmosphere. The movement of water takes place because there is a water potential gradi- ent from relatively high soil water potential to successively lower water potentials in roots, stems, leaves, and the atmosphere.
| Osmosis occurs according to the concentration gradient of water across the membrane, which is inversely proportional to the concentration of solutes.Osmosis occurs until the concentration gradient of water goes to zero or until the hydrostatic pressure of the water balances the osmotic pressure.Osmosis occurs when there is a concentration gradient of a solute within a solution, but the membrane does not allow diffusion of the solute. |

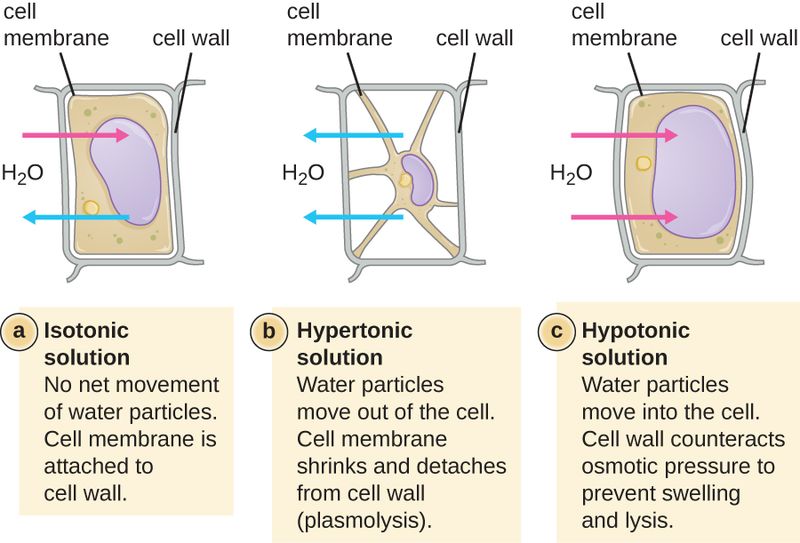
Plasmolysis
If you place turgid carrot and celery sticks in a 10% solution of salt in water, they soon lose their rigidity and become limp enough to curl around your finger. The water potential inside the carrot cells is greater than the water potential outside, and so diffusion of water out of the cells into the salt solution takes place. If you were to examine such cells with a microscope, you would see that the vacuoles, which are largely water, had disappeared and that the cytoplasm was clumped in the middle of the cell, having shrunken away from the walls. Such cells are said to be plasmolyzed. This loss of water through osmosis, which is accompanied by the shrinkage of protoplasm away from the cell wall, is called plasmolysis . If plasmo- lyzed cells are placed in fresh water before permanent damage is done, water reenters the cell by osmosis, and the cells be- come turgid once more.
| A hypertonic solution contains a higher concentration of solutes compared to another solution. The opposite solution with a lower concentration is known as the hypotonic solution. Generally, plants prefer to live in hypotonic environments. In a hypotonic environment, water easily floods plant cells and they can remain turgid, or rigid, due to pressures exerted on their cell walls by the influx of water. The plants use this water potential to give their bodies structure and move water from the roots to the top of the plant. However, many plants have adapted to live in hypertonic environments. Marshes by the sea, mangrove swamps, and other brackish waters contain a much higher salt content than fresh water. The soil becomes saturated with these salts, creating a much higher solute concentration in the soil. Passive diffusion is the movement of molecules from a higher concentration to a lower concentration. Osmosis and plasmolysis are two events that occur due to the movement of water molecules. Water is considered as the universal solvent in cells that dissolves polar molecules. The main difference between osmosis and plasmolysis is that osmosis is the movement of water molecules from high water potential to a lower water potential across a semipermeable membrane whereas plasmolysis is the shrinkage of a cell due to the persisting movement of the water molecules out of the cell. Plasma membrane serves as the semipermeable membrane during osmosis. The two types of osmosis are endosmosis and exosmosis. Plasmolysis occurs due to persisting exosmosis. |
Imbibition
Osmosis is not the only force involved in the absorption of water by plants. Colloidal materials (i.e., materials that contain a permanent suspension of fine particles) and large molecules, such as cellulose and starch, usually develop electrical charges when they are wet. The charged colloids and molecules attract water molecules, which adhere to the internal surfaces of the materials. Because water molecules are polar, they can become both highly adhesive to large organic molecules such as cellu- lose and cohesive with one another. As discussed, polar molecules have slightly different electrical charges at each end due to their asymmetry. This process, known as imbibition, results in the swelling of tissues, whether they are alive or dead, often to several times their original volume. Imbibition is the initial step in the germination of seeds.
Active Transport
Most molecules needed by cells are polar, and those of solutes may set up an electrical gradient across a semi-permeable membrane of a living cell. To pass through the membrane, molecules require special embedded transport proteins (see Fig. 3.7). The transport proteins are believed to occur in two forms: one facilitating the transport of specific ions to the outside of the cell and the other facilitating the transport of specific ions into the cell.
The plants absorb and retain these solutes against a diffusion (or electrical) gradient through the expenditure of energy. This process is called active transport. The precise mechanism of active transport is not fully understood, but recent evi- dence suggests that this process involves an enzyme complex and what has been referred to as a proton pump. The pump in- volves the plasma membrane of plant and fungal cells and sodium and potassium ions in animal cells. Both pumps are ener- gized by special energy-storing ATP molecules.
Mangroves, saltbush, and certain algae thrive in areas where the water or soil contains enough salt to kill most vegeta- tion. Such plants accumulate large amounts of organic solutes, including the carbohydrate mannitol and the amino acid proline. The organic solutes facilitate osmosis, despite the otherwise adverse environment. The leaves of some mangroves also have salt glands through which they excrete excess salt.
WATER AND ITS MOVEMENT THROUGH THE PLANT
Why do living plants require so much water? Water constitutes about 90% of the weight of young cells. The thousands of enzyme actions and other chemical activities of cells take place in water, and additional, although relatively negligible, amounts are used in the process of photosynthesis. The exposed surfaces of the mesophyll cells within the leaf have to be moist at all times, for it is through this film of water that the carbon dioxide molecules needed for the process of photosynthe- sis enter the cell from the air. Water is also needed for cell turgor, which gives rigidity to herbaceous plants.
Consider also what it must be like in the mesophyll of a flattened leaf that is fully exposed to the midsummer sun in areas where the air temperature soars to well over 38°C (100°F) in the shade. If it were not for the evaporation of water molecules from the moist surfaces, which brings about cooling, and reradiation of energy by the leaf, the intense heat could damage the plant. Sometimes, the transpiration is so rapid that more water is lost than is taken in. The stomata may then close, preventing wilting. The relation and role of abscisic acid in excessive water loss is discussed elsewhere .
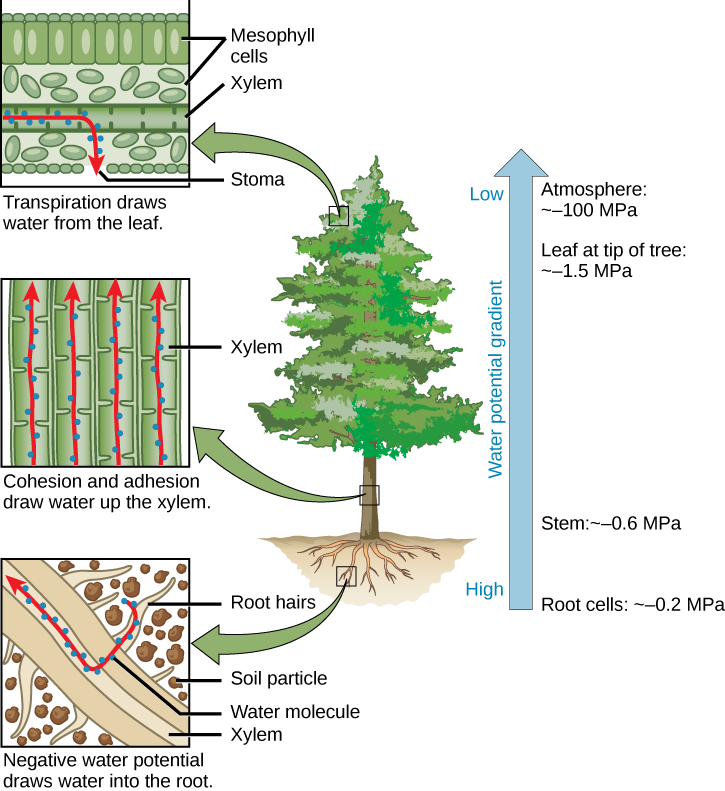
How does water travel through the roots from 3 to 6 meters (10 to 20 feet) or more beneath the surface and then up the trunk to the topmost leaves of a tree that is more than 90 meters (300 feet) tall? We know that interconnected tubes of xylem extend throughout the plant, from the young roots up through the stem and branches to the tiny veinlets of the leaves. We also know that the water, following a water potential gradient, gets to the start of this “plumbing system” by osmosis. Water is then raised through the columns apparently by a combination of factors, and the process has been the subject of much debate for the past 200 years
The Cohesion-Tension Theory
Water molecules move partly through cell cytoplasm and partly through spaces between cells; they also move between cellulose fibers in the walls and through spaces in the centers of dead cells. Most water and solutes can travel across the epi- dermis and cortex via the cell walls until they reach the endodermis. There, the water and solutes are forced by Casparian strips to cross the cytoplasm of the endodermal cells on their way to the vessels or tracheids of the xylem.
If significant transpiration is occurring, the roots are likely to grow rapidly toward available water. In corn plants, for ex- ample, the main roots may grow at a rate of more than 6 centimeters (2.3 inches) a day. Solutes, as well as water, may move so rapidly during periods of rapid transpiration that there is little osmosis taking place across the endodermis. Scientists be- lieve that at such times water may be pulled through the roots by bulk flow, which is the passive movement of a liquid from higher to lower water potential.
In summary: “columns” of water molecules are pulled through the plant from roots to leaves, and the abundant water of a normally moist soil supplies these “columns” as the water continues to enter the root by osmosis (see Fig. 9.10); simply put, the difference between the water potentials (water “concentrations”) of two areas (e.g., soil and the air around stomata) generates the force to transport water in a plant.
| Figure: -cylinderThe cohesion and adhesion of water in the vessel element helps water move up the vessel without breaking under tension. Adhesion occurs when water molecules are attracted to the walls of the vessel element, which has thick walls with lignin, a stiff substance. Cohesion occurs when water molecules are attracted to each other. This is due to hydrogen bonds, which form between the partially negative oxygen of one molecule and the partially positive hydrogen of another molecule. Hydrogen bonds are a strong intermolecular force. As some water molecules move up the vessel element, they pull other water molecules with them. Water molecules move up the xylem (in one direction). Figure-tree: The cohesion–tension theory is shown. Evaporation from the mesophyll cells produces a negative water potential gradient that causes water to move upwards from the roots through the xylem. Water potential is measured in megapascals (MPa), which is a measure of pressure. Water potential decreases from the root cells (-0.2 MPa) to the stem (-0.6 MPa) to the leaf at the tip of a tree (-1.5 MPa) to the atmosphere (-100 MPa). Transpiration draws water from the leaf. The water leaves the tube-shaped xylem and enters the air space between mesophyll cells. Finally, it exits through the stoma. Cohesion and adhesion draw water up the xylem. Negative water potential draws water into the root. Water moves from the spaces between soil particles into the root hairs, and into the xylem of the root. |

REGULATION OF TRANSPIRATION
Two guard cells and an opening called the stoma (plural: stomata) comprise the stomatal apparatus. These stomatal appara- tuses, which often occupy 1% or more of the surface area of a leaf, regulate transpiration and gas exchange. Control of tran- spiration is, however, strongly influenced by the water-vapor concentration of the atmo-sphere. The guard cells bordering each stoma have relatively elastic walls with radially oriented microfibrils, making them analogous to pairs of sausage- shaped balloons joined at each end, each with a row of rubber bands around it. The part of the wall adjacent to the hole itself is considerably thicker than the remainder of the wall . This thickness allows each stoma to be opened and closed by means of changes in the turgor of the guard cells. The stoma is closed when turgor pressure is low and open when turgor pressure is high. Changes in turgor pressures in the guard cells, which contain chloroplasts, take place when they are exposed to changes in light intensity, carbon dioxide concentration, or water concentration.
Changes in turgor pressure take place when osmosis and active transport between the guard cells and other epidermal cells bring about shifts in solute concentrations. While photosynthesis is occurring in the guard cells, they expend energy to acquire potassium ions from adjacent epidermal cells, leading to the opening of the stomata. When photosynthesis is not oc- curring in the guard cells, the potassium ions leave, and the stomata close. With an increase in potassium ions, the water po- tential in the guard cells is lowered, and the osmosis that takes place as a result brings in water that makes the cells turgid. The departure of potassium ions also results in water leaving, making the cells less turgid and causing the stomata to close (see Fig. 9.13).
Stomata will close passively whenever water stress occurs, but there is evidence that the hormone abscisic acid is pro- duced in leaves subject to water stress and that this hormone causes membrane leakages, which induce a loss of potassium ions from the guard cells and cause them to deflate.
The stomata of most plants are open during the day and closed at night. However, the stomata of a number of desert plants are open only at night when there is less water stress on the plants. This conserves water but makes carbon dioxide needed for photosynthesis inaccessible during the day. Such plants convert the carbon dioxide available at night to organic acids, which are stored in cell vacuoles. The organic acids are then converted back to carbon dioxide during the day when photosynthesis occurs . A specialized form of photosynthesis called CAM photosynthesis uses the carbon dioxide released from the organic acids. CAM photosynthesis is discussed .
Other desert plants have their stomata recessed below the surface of the leaf or stem in small chambers. These chambers, called stomatal crypts, often are partially filled with epidermal hairs, which further reduce water loss by slowing down air movement. Similar recessed stomata are found in the leaves of pine trees, which have little water available to them in winter when the soil is frozen (see Fig. 7.12). A few tropical plants that occur in damp, humid areas (e.g., ruellias; see also Fig. 4.13B) have stomata that are raised above the surface of the leaf, while plants of wet habitats generally lack stomata on submerged surfaces.
Although light and carbon dioxide concentration affect transpiration rates, several other factors play at least an indirect role. For example, air currents speed up transpiration as they sweep away water molecules emerging from stomata. Humidity plays an inverse but direct role in transpiration rates: high humidity reduces transpiration, and low humidity accelerates it. Temperature also plays a role in the movement of water molecules out of a leaf. The transpiration rate of a leaf at 30°C (86°F), for example, is about twice as great as it is for the same leaf at 20°C (68°F). The various adaptive modifica- tions of leaves and their surfaces and the availability of water to the roots also may play important roles in influencing the amount of water transpired. Leaf modifications are discussed.
If a cool night follows a warm, humid day, water droplets may be produced through structures called hydathodes at the tips of veins of the leaves of some herbaceous plants. This loss of water in liquid form is called guttation. Miner- als absorbed at night are pumped into the intercellular spaces surrounding the vessels and tracheids of the xylem. As a result, the water potential of the xylem elements is lowered, and water moves into them from the surrounding cells. In the absence of transpiration at night, the pressure in the xylem elements builds to the point of forcing liquid water out of the hydathodes in the leaves. Although the droplets resemble dew, the two should not be confused. Dew is water that is condensed from the air, while guttation water is literally forced out of the plant by root pressure. As the sun strikes the droplets in the morning, they dry up, leaving a residue of salts and organic substances, one of which is used in the manufacture of commercial flavor enhancers (e.g., the monosodium glutamate in products such as Accent®). In the tropics, the amount of water produced by guttation can be considerable. In taro plants, used by the Polynesians to make poi, a single leaf may overnight produce as much as a cupful (about 240 milliliters) of water through guttation.
TRANSPORT OF FOOD SUBSTANCES (ORGANIC SOLUTES) IN SOLUTION
One of the most important functions of water in the plant involves the translocation (transportation) of food substances in solution by the phloem, a process that has only recently come to be better understood. Many of the studies that led to our pre- sent knowledge of the subject used aphids (small, sucking insects) and organic compounds designed as radioactive tracers.
Most aphids feed on phloem by inserting their tiny, tubelike mouthparts (stylets) through the leaf or stem tissues until a sieve tube is reached and punctured. The turgor pressure of the sieve tube then forces the fluid present in the tube through the aphid’s digestive tract, and it emerges at the rear as a droplet of “honeydew.” In some studies, research workers anesthetized feeding aphids and cut their stylets so that much of the tiny tube remained where it had been inserted. Fluid exuded (some- times for many hours) from the cut stylets and was then collected and analyzed .
Carbon dioxide, a basic raw material of photosynthesis, can be synthesized with radioactive carbon. By exposing a pho- tosynthesizing leaf to radioactive carbon dioxide, the pathway of manufactured food substances can be traced. The radioac- tive substances produce on photographic film an image corresponding to the food pathway. Data obtained from such studies reveal that food substances in solution are confined entirely to the sieve tubes while they are being transported. At one time, it was believed that ordinary diffusion and cyclosis were responsible for the movement of the sub- stances from one sieve tube member to the next, but it is now known that the substances move through the phloem at ap- proximately 100 centimeters (almost 40 inches) per hour—far too rapid a movement to be accounted for by diffusion and cyclosis alone.
Summary
- Molecules and ions are in constant random motion and tend to distribute themselves evenly in the space available to them. They move from a region of higher concentration to a region of lower concentration by simple diffusion along a diffusion gradient; they may also move against a diffusion gradient. Evenly distributed molecules are in a state of equi- librium. Diffusion rates are affected by temperature, molecule size and density, and other factors.
- Osmosis is the diffusion of water through a semipermeable membrane. It takes place in response to concentration differ- ences of dissolved substances.
- Osmotic pressure or potential is the pressure required to prevent osmosis from taking place. The pressure that develops in a cell as a result of water entering it is called turgor. Water moves from a region of higher water potential (osmotic po- tential and pressure potential combined) to a region of lower water potential when osmosis is occurring. Osmosis is the primary means by which plants obtain water from their environment.
- Plasmolysis is the shrinkage of the cytoplasm away from the cell wall as a result of osmosis taking place when the water potential inside the cell is greater than outside.
- Imbibition is the attraction and adhesion of water molecules to the internal surfaces of materials; it results in swelling and is the initial step in the germination of seeds.
- Active transport is the expenditure of energy by a cell that results in molecules or ions entering or leaving the cell against a diffusion gradient.
- Water that enters a plant passes through it and mostly transpires into the atmosphere via stomata. Water retained by the plant is used in photosynthesis and other metabolic activities.
- The cohesion-tension theory postulates that water rises through plants because of the adhesion of water molecules to the walls of the capillary-conducting elements of the xylem, cohesion of the water molecules, and tension on the water col- umns created by the pull developed by transpiration.
- The translocation of food substances takes place in a water solution, and according to the pressure-flow hypothesis, such substances flow along concentration gradients between their sources and sinks.
- Transpiration is regulated by humidity and the stomata, which open and close through changes in turgor pressure of the guard cells. These changes, which involve potassium ions, result from osmosis and active transport between the guard cells and the adjacent epidermal cells.
- Aquatic, desert, tropical, and some cold-zone plants have modifications of stomatal apparatuses or specialized forms of photosynthesis that adapt them to their particular environments.
- Guttation is the loss of water at night in liquid form through hydathodes at the tips of leaf veins.
Growth phenomena are controlled by both internal and external means and by chemical and physical forces in balance with one another. Besides carbon, hydrogen, and oxygen, 15 other elements are essential to most plants. When any of the essential elements are deficient in the plant, characteristic deficiency symptoms appear
The Pressure-Flow Hypothesis
At present, the most widely accepted theory for movement of substances in the phloem is called the pressure-flow (or mass- flow) hypothesis. According to this theory, food substances in solution (organic solutes) flow from a source, where water en- ters by osmosis (e.g., a food-storage tissue, such as the cortex of a root or rhizome, or a food-producing tissue, such as the mesophyll tissue of a leaf). The water exits at a sink, which is a place where food is utilized, such as the growing tip of a stem or root. Food substances in solution (organic solutes) are moved along concentration gradients between sources and sinks.
First, in a process called phloem-loading, sugar, by means of active transport, enters the sieve tubes of the smallest veinlets. This decreases the water potential in the sieve tubes, and water then enters these phloem cells by osmosis. Tur- gor pressure, which develops as this osmosis occurs, is responsible for driving the fluid through the sieve-tube network toward the sinks.
As the food substances (largely sucrose) in solution are actively removed at the sink, water also exits the sink ends of sieve tubes, and the pressure in these sieve tubes is lowered, causing a mass flow from the higher pressure at the source to the lower pressure at the sink. Most of the water diffuses back to the xylem, where it then returns to the source and is transpired or recirculated. The pressure-flow hypothesis explains how nontoxic dyes applied to leaves or substances entering the sieve tubes, such as viruses introduced by aphids, are carried through the phloem.
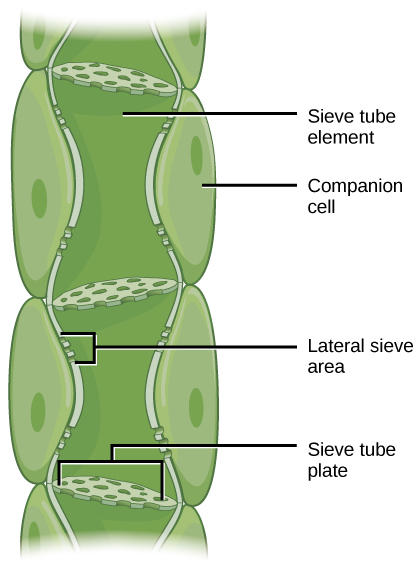
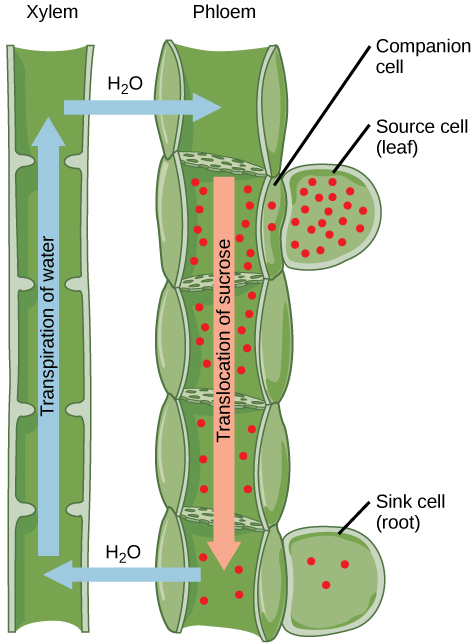
| Translocation: Transport from Source to Sink Photosynthates, such as sucrose, are produced in the mesophyll cells of photosynthesizing leaves. From there they are translocated through the phloem to where they are used or stored. Mesophyll cells are connected by cytoplasmic channels called plasmodesmata. Photosynthates move through these channels to reach phloem sieve-tube elements (STEs) in the vascular bundles. From the mesophyll cells, the photosynthates are loaded into the phloem STEs. The sucrose is actively transported against its concentration gradient (a process requiring ATP) into the phloem cells using the electrochemical potential of the proton gradient. This is coupled to the uptake of sucrose with a carrier protein called the sucrose-H+ symporter. Phloem STEs have reduced cytoplasmic contents, and are connected by a sieve plate with pores that allow for pressure-driven bulk flow, or translocation, of phloem sap. Companion cells are associated with STEs. They assist with metabolic activities and produce energy for the STEs. Figure 19.6.6: Phloem is comprised of cells called sieve-tube elements. Phloem sap travels through perforations called sieve tube plates. Neighboring companion cells carry out metabolic functions for the sieve-tube elements and provide them with energy. Lateral sieve areas connect the sieve-tube elements to the companion cells. ——— Once in the phloem, the photosynthates are translocated to the closest sink. Phloem sap is an aqueous solution that contains up to 30 percent sugar, minerals, amino acids, and plant growth regulators. The high percentage of sugar decreases Ψs, which decreases the total water potential and causes water to move by osmosis from the adjacent xylem into the phloem tubes, thereby increasing pressure. This increase in total water potential causes the bulk flow of phloem from source to sink (Figure 19.6.7). Sucrose concentration in the sink cells is lower than in the phloem STEs because the sink sucrose has been metabolized for growth, or converted to starch for storage or other polymers, such as cellulose, for structural integrity. Unloading at the sink end of the phloem tube occurs by either diffusion or active transport of sucrose molecules from an area of high concentration to one of low concentration. Water diffuses from the phloem by osmosis and is then transpired or recycled via the xylem back into the phloem sap. Figure 19.6.7: Sucrose is actively transported from source cells into companion cells and then into the sieve-tube elements. This reduces the water potential, which causes water to enter the phloem from the xylem. The resulting positive pressure forces the sucrose-water mixture down toward the roots, where sucrose is unloaded. Transpiration causes water to return to the leaves through the xylem vessels. |
