The use of energy from sunlight by photosynthesis is the basis of life on earth
Chlorophyll is the main photosynthetic pigment
Pigments capture energy from sunlight
The energy content of light depends on its wavelength
In Berlin at the beginning of the twentieth century Max Planck and Albert Einstein, two Nobel Prize winners, carried outthe epoch-making studies proving
that light has a dual nature. It can be regarded as an electromagnetic wave as well as an emission of particles, which are termed light quanta or photons.
The energy of the photon is proportional to its frequency v:
where h is the Planck constant (6.6 · 1034 J s) and c the velocity of the light (3 · 108 m s1). is the wavelength of light.
The mole (abbreviated to mol) is used as a chemical measure for the amount of molecules and the amount of photons corresponding to 6 · 1023 molecules or photons (Avogadro number NA). The energy of one mol photons amounts to:
In order to utilize the energy of a photon in a thermodynamic sense, this energy must be at least as high as the Gibbs free energy of the photochemi- cal reaction involved. (In fact much energy is lost during energy conversion (section 3.4), with the consequence that the energy of the photon must be higher than the Gibbs free energy of the corresponding reaction.) We can equate the Gibbs free energy G with the energy of the absorbed light:
The introduction of numerical values of the constants h, c, and NA yields:
The human eye perceives only the small range between about 400 and 700 nm of the broad spectrum of electromagnetic waves (Fig. 2.2). The light in this range, where the intensity of solar radiation is especially high, is uti- lized in plant photosynthesis. Bacterial photosynthesis, however, is able to utilize light in the infrared range.
According to equation 2.3 the energy of irradiated light is inversely proportional to the wavelength. Table 2.1 shows the light energy per mol photons for light of different colors. Consequently, violet light has an energy of about 300 kJ/mol photons. Dark blue light, with the highest wavelength (700 nm) that can still be utilized by plant photosynthesis, contains 170 kJ/ mol photons. This is only about half the energy content of violet light.
In photosynthesis of a green plant, light is collected primarily by chlorophylls, pigments that absorb light at a wavelength below 480 nm and between 550 and 700 nm (Fig. 2.3). When white sunlight falls on a chlorophyll layer, the green light with a wavelength between 480 and 550 nm is not absorbed, but is reflected. This is why plant chlorophylls and whole leaves appear green.
Experiments carried out between 1905 and 1913 in Zurich and Berlin by Richard Willstätter and his collaborators led to the discovery of the structural formula of the green leaf pigment chlorophyll, a milestone in the history of chemistry. This discovery made such an impact that Richard Willstätter was awarded the Nobel Prize in Chemistry as early as 1915. There are different classes of chlorophylls. Figure 2.4 shows the structural formulas of chlorophyll-a and chlorophyll-b (chl-a, chl-b). The basic struc- ture is a ring made of four pyrroles, a tetrapyrrole, which is also named porphyrin. Mg is present in the center of the ring as the central atom. Mg is covalently bound with two N atoms and coordinately bound to the other two atoms of the tetrapyrrole ring. A cyclopentanone is attached to ring c. At ring d a propionic acid group forms an ester with the alcohol phytol. Phytol consists of a long branched hydrocarbon chain with one C-C double bond. It is derived from an isoprenoid, formed from four isoprene units (section 17.7). This long hydrophobic hydrocarbon tail renders the chlorophyll highly soluble in lipids and therefore promotes its presence in the membrane phase. Chlorophyll always occurs bound to proteins. Chl-b contains a formyl residue in ring b instead of the methyl residue as in chl-a. This small difference has a large influence on light absorption. Figure 2.3 shows that the absorption spectra of chl-a and chl-b differ markedly.
In plants, the ratio chl-a to chl-b is about three to one. Only chl-a is a constituent of the photosynthetic reaction centers (sections 3.6 and 3.8) and therefore it can be regarded as the central photosynthesis pigment. In a wide range of the visible spectrum, however, chl-a does not absorb light (Fig. 2.3). This non-absorbing region is named the “green window.” The absorption gap is narrowed by the light absorption of chl-b, with its first maximum at a higher wavelength than chl-a and the second maximum at a lower wavelength. As shown in section 2.4, the light energy absorbed by chl-b can be transferred very efficiently to chl-a. In this way, chl-b enhances the plant’s efficiency for utilizing sunlight energy.
The structure of chlorophylls has remained remarkably constant during the course of evolution. Purple bacteria, probably formed more than 3 billion years ago, contain as photosynthetic pigment a bacteriochlorophyll-a, which differs from the chl-a shown in Fig. 2.4 only by the alteration of one side chain and by the lack of one double bond. This, however, influences light absorption; both absorption maxima are shifted outwards and the non-absorbing spectral region in the middle is broadened. This shift allows purple bacteria to utilize light in the infrared region.
The tetrapyrrole ring not only is a constituent of chlorophyll but also has attained a variety of other functions during evolution. It is involved in methane formation by bacteria with Ni as the central atom. With Co it forms cobalamin (vitamin B12), which participates as a cofactor in reac- tions in which hydrogen and organic groups change their position. With Fe instead of Mg as the central atom, the tetrapyrrole ring forms the basic structure of hemes (Fig. 3.24), which as cytochromes function as redox carriers in electron transport processes (sections 3.7 and 5.5) and as myoglobin or hemoglobin stores or transports oxygen in aerobic organ- isms. The tetrapyrrole ring in animal hemoglobin differs only slightly from the tetrapyrrole ring of chl-a (Fig. 2.4).
It seems remarkable that a substance that attained a certain function during evolution is being utilized after only minor changes for completely different functions. The reason for this functional variability is that the reactivity of compounds such as chlorophyll or heme is governed to a great extent by the proteins to which they are bound.
Chlorophyll molecules are bound to chlorophyll-binding proteins. In a complex with proteins the absorption spectrum of the bound chlorophyll differs considerably from the absorption spectrum of the free chlorophyll. The same applies for other light-absorbing compounds, such as carotenoids, xanthophylls, and phycobilins, which also occur bound to proteins. These complexes will be discussed in the following sections. For better discrimi- nation in this text book, free absorbing compounds are called chromophore (Greek, carrier of color) and the chromophore-protein complexes are called pigments. Pigments are further characterized by the wavelength of their absorption maximum. Chlorophyll-a700 describes a pigment of protein-chl-a complex with an absorption maximum of 700 nm. Another common desig- nation is P700; this nomination leaves the nature of the chromophore open.
Light absorption excites the chlorophyll molecule
What happens when a chromophore absorbs a photon? When a photon with a certain wavelength hits a chromophore molecule that absorbs light
of this wavelength, the energy of the photon excites electrons and trans- fers them to a higher energy level. This occurs as an “all or nothing” proc- ess. According to the principle of energy conservation expressed by the first law of thermodynamics, the energy of the chromophore is increased by the energy of the photon, which results in an excited state of the chromo- phore molecule. The energy is absorbed only in discrete quanta, resulting in discrete excitation states. The energy required to excite a chromophore molecule depends on the chromophore structure. A general property of chromophores is that they contain many conjugated double bonds, 10 in the case of the tetrapyrrole ring of chl-a. These double bonds are delocalized. Figure 2.5 shows two possible resonance forms.
After absorption of energy, an electron of the conjugated system is ele- vated to a higher orbit. This excitation state is termed singlet. Figure 2.6 shows a scheme of the excitation process. As a rule, the higher the number of double bonds in the conjugated system, the lower the amount of energy required to produce a first singlet state. For the excitation of chlorophyll, dark red light is sufficient, whereas butadiene, with only two conjugated double bonds, requires energy-rich ultraviolet light for excitation. The light absorption of the conjugated system of the tetrapyrrole ring is influenced by the side chains. Thus, the differences in the absorption maxima of chl-a and chl-b mentioned previously can be explained by an electron attracting effect of the carbonyl side chain in ring b of chl-b (Fig. 2.5).
The spectra of chl-a and chl-b (Fig. 2.3) each have two main absorption maxima, showing that each chlorophyll has two main excitation states. In addition, chlorophylls have minor absorption maxima, which for the sake of simplicity will not be discussed here. The two main excitation states of chlo- rophyll are known as the first and second singlet (Fig. 2.6). The absorption
maxima in the spectra are relatively broad. At a higher resolution the spec- tra can be shown to consist of many separate absorption lines. This fine structure of the absorption spectra is due to chlorophyll molecules that are in the ground and in the singlet states as well in rotation and vibration. In the energy scheme the various rotation and vibration energy levels are drawn as fine lines and the corresponding ground states as solid lines (Fig. 2.6).
The energy levels of the various rotation and vibration states of the ground state overlap with the lowest energy levels of the first singlet. Analogously, the energy levels of the first and the second singlet also over- lap. If a chlorophyll molecule absorbs light in the region of its absorption maximum (blue light), one of its electrons is elevated to the second singlet state. This second singlet state with a half-life of only 1012 s is too unsta- ble to use its energy for chemical work. The excited molecules lose energy as heat by rotations and vibrations until the first singlet state is reached. This first singlet state can also be attained by absorption of a photon of red light, which contains less energy. The first singlet state is much more stable than the second one; its half-life time is 4 · 109 s.
The return of the chlorophyll molecule from the first singlet state to the ground state can proceed in different ways:
The most important path for the conversion of the energy released upon the return of the first singlet state to the ground state is its utiliza- tion for chemical work. The chlorophyll molecule transfers the excited electron from the first singlet state to an electron acceptor and a pos- itively charged chlorophyll radical chl remains. This is possible since the excited electron is bound less strongly to the chromophore molecule than in the ground state. Section 3.5 describes in detail how the elec- tron can be transferred back from the acceptor to the chl radical via an electron transport chain. When the chlorophyll molecule returns to the ground state, the free energy derived from this process is conserved for chemical work. As an alternative, the electron deficit in the chl radical may be replenished by another electron donor (e.g., water (section 3.6)).
The excited chlorophyll can return to the ground state by releasing exci- tation energy as light; this emitted light is named fluorescence. Due to vibrations and rotations, part of the excitation energy is usually lost as heat, with the result that the fluorescence light has less energy (corre- sponding to a longer wavelength) than the energy of the excitation light, which was required for attaining the first singlet state (Fig. 2.7).
It is also possible that the return from the first singlet to the ground state proceeds in a stepwise fashion via the various levels of vibration and rotation energy, by which the energy difference is completely con- verted into heat.
By releasing part of the excitation energy as heat, the chlorophyll mol- ecule can attain a lower energy excitation state, called the first triplet state. This triplet state cannot be reached directly from the ground state by excitation, since the spin of the excited electrons has been reversed. Since the probability of a reversal spin is low, the triplet state does not occur frequently. In the case of a very high excitation, however, some of the electrons of the chlorophyll molecules can reach this state. By emitting so-called phosphorescent light, the molecule can return from the triplet state to the ground state. Phosphorescent light is lower in energy than the light required to attain the first singlet state. The return from the triplet state to the ground state requires a reversal of the electron spin. As this is rather improbable, the triplet state, in comparison to the first singlet state, has a relatively long half-life time (104 to 102 s). The triplet state of the chlorophyll has no function in photosynthesis per se. In its triplet state, however, the chlorophyll can excite oxygen to a sin- glet state, whereby the oxygen becomes very reactive (reactive oxygen species, ROS, section 5.7) with a damaging effect on cell constituents. Section 3.10 describes how the plant manages to protect itself from the harmful singlet oxygen.
The return to the ground state can be coupled with the excitation of a neighboring chromophore molecule. This transfer is important for the function of the antennae and will be described in the following section.

Figure 2.2 Spectrum of the electromagnetic radiation. The enlargement in red illustrates the visible spectrum.

Figure 2.3 Absorption spectrum of chlorophyll-a (chl-a), chlorophyll-b (chl-b) and of the xanthophyll lutein dissolved in acetone. The intensity of the sun’s radiation at different wavelengths is given as a comparison
Video- Excitation of Chlorophyll by Light
Video- The Chloroplast
Table 2.1: The energy content and the electrochemical potential difference of photons of different wavelengths
| Wavelengths (nm) | Light color | Energy content kJ/mol photons | E e volt |
| 700 | Red | 170 | 1.76 |
| 650 | Bright red | 183 | 1.90 |
| 600 | Yellow | 199 | 2.06 |
| 500 | Blue green | 238 | 2.47 |
| 440 | Blue | 271 | 2.80 |
| 400 | Violet | 298 | 3.09 |

Figure 2.4 Structural formula of chlorophyll-a. In chlorophyll-b the methyl group in ring b is replaced by a formyl group (A). The phytol side chain in red gives chlorophyll a lipid character.
Video- Emission and Absorption Spectra
Video- Light Absorption, Reflection, and Transmission
Video- Light-Harvesting: Photosynthetic Pigments and Exciton Transfer (Playlist photosynthesis)
Video- Light-Harvesting: The Antenna Complex
BREAK

Figure 2.5 Resonance structures of chlorophyll-a. In the region marked red, the double bonds are not localized; the electrons are distributed over the entire conjugated system. The formyl residue of chlorophyll-b attracts electrons and thus affects the electrons of the conjugated system.

Figure 2.6 Schematic presentation of the excitation states of chlorophyll-a and their return to the ground state. The released excitation energy is converted
An antenna is required to capture light
In order to excite a photosynthetic reaction center, a photon with defined energy content has to react with a chlorophyll molecule in the reaction center. The probability is very low that a photon not only has the proper energy, but also hits the pigment exactly at the site of the chlorophyll mol- ecule. Therefore efficient photosynthesis is possible only when the energy of photons of various wavelengths is captured over a certain surface by a so-called antenna (Fig. 2.8). Similarly, radio and television sets could not work without an antenna.
The antennae of plants consist of a large number of protein-bound chlorophyll molecules that absorb photons and transfer their energy to the reaction center. Only a few thousandths of the chlorophyll molecules in the leaf are constituents of the actual reaction centers; the remainder are contained in the antennae. Observations made as early as 1932 by Robert Emerson and William Arnold in the United States indicated that the large majority of chlorophyll molecules are not part of the reaction centers. The two researchers illuminated a suspension of the green alga Chlorella with light pulses of 10 s duration, interrupted by dark intervals of 20 ms. Evolution of oxygen was used as a measure for photosynthesis. The light pulses were made so short that chlorophyll could undergo only one photo- synthetic excitation cycle and a high light intensity was chosen in order to achieve maximum oxygen evolution. Apparently the photosynthetic appa- ratus was thus saturated with photons. Analysis of the chlorophyll content of the algae suspension showed that under saturating conditions only one molecule of O2 was formed per 2,400 chlorophyll molecules.
In the following years Robert Emerson refined these experiments and
was able to show when pulses were applied at very low light intensity, the amount of oxygen formed increased proportionally with the light intensity. From this it was calculated that the release of one molecule of oxygen had a minimum quantum requirement of about eight photons. These results set- tled a long scientific dispute with Otto Warburg, who had concluded from his experiments that only four photons are required for the evolution of one molecule of O2. Later it was recognized that each of the two reaction cent- ers requires four photons for the formation of O2. Moreover, the results of Emerson and Arnold allowed the calculation that about 300 chlorophyll molecules are associated with one reaction center. These are constituents of the antennae. The antennae contain additional accessory pigments to utilize those photons where the wavelength corresponds to the “green window” between the absorption maxima of the chlorophylls. In higher plants these pigments are carotenoids, mainly xanthophylls, including lutein and the related vio- laxanthin as well as carotenes such as -carotene to name the major com- pound (Fig. 2.9). Moreover, an important function of these carotenoids in the antennae is to prevent the formation of the harmful triplet state of the chlorophylls (section 3.10). Important constituents of the antennae in cyanobacteria are phycobilins, which will be discussed at the end of this chapter.
How is the excitation energy of the photons captured in the antennae and transferred to the reaction centers?
The transfer of energy in the antennae via electron transport from chromo- phore to chromophore in a sequence of redox processes, as in the elec- tron transport chains of photosynthesis or of mitochondrial respiration (Chapters 3 and 5), could be excluded, since such an electron transport would need considerable activation energy. This is not the case, since a flux of excitation energy can be measured in the antennae at temperatures as low as 1 K. At these low temperatures light absorption and fluorescence still occur, whereas chemical processes catalyzed by enzymes are completely inactive. Under these conditions the energy transfer in the antennae pro- ceeds according to a mechanism that is related to those of light absorption and fluorescence.
When chromophores are positioned very close to each other, the quan- tum energy of an irradiated photon is transferred from one chromophore to the next. One quantum of light energy is named a photon, one quantum of excitation energy transferred from one molecule to the next is termed an exciton. A prerequisite for the transfer of excitons is a specific position- ing of the chromophores. This is arranged by proteins, and therefore the chromophores of the antennae always occur as protein complexes.
The antennae of plants consist of an inner part and an outer part (Fig. 2.10). The outer antenna, formed by the light harvesting complexes (LHCs), col- lects the light. The inner antenna, consisting of the core complexes, is an integral constituent of the reaction centers; it also collects light and con- ducts the excitons that were collected in the outer antenna to the photosyn- thetic reaction centers.
The LHCs are composed of polypeptides, which bind chl-a, chl-b, xan- thophylls, and carotenes. These proteins, termed LHC polypeptides, are encoded in the nucleus. A plant contains many different LHC polypeptides. In a tomato, for instance, at least 19 different genes for LHC polypeptides have been found, which are very similar to each other and are members of a multigene family. They are homologous, as they have all evolved from a common ancestor.
Plants contain two reaction centers, which are arranged in sequence: a reaction center of photosystem II (PS II), which has an absorption maxi- mum at 680 nm, and a photosystem I (PS I) with an absorption maximum at 700 nm. The function of these reaction centers will be described in sec- tions 3.6 and 3.8. Both photosystems are composed of different LHCs.
The function of an antenna is illustrated by the antenna of photosystem II
The antenna of the PS II reaction center contains primarily four LHCs termed LHC-IIa–d. The main component is LHC-IIb; it represents 67% of

Figure 2.8 Photons are collected by an antenna and their energy is transferred to the reaction center. In this scheme the squares represent chlorophyll molecules. The excitons conducted to the reaction center cause a charge separation (section
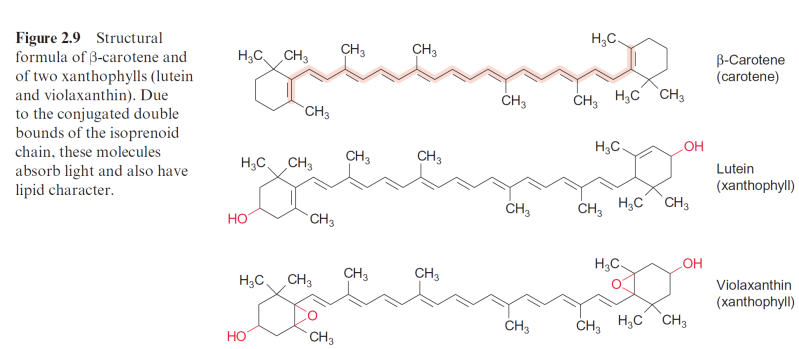
Figure 2.9 Structural formula of -carotene and of two xanthophylls (lutein
vcideo- 3 Major Classes of Pigments in Photosynthesis
video-Photosynthetic Electron Transport
Oxidation and reduction in cellular respiration | Biology | Khan Academy
Photosynthesis and Respiration
Photosynthesis
Z scheme/light reaction of photosynthesis with reduction potential
Redox Reactions and the light reactions of photosynthesis– 5.7 – Biol 189

Figure 2.10 Schematic presentation of a higher plant antenna
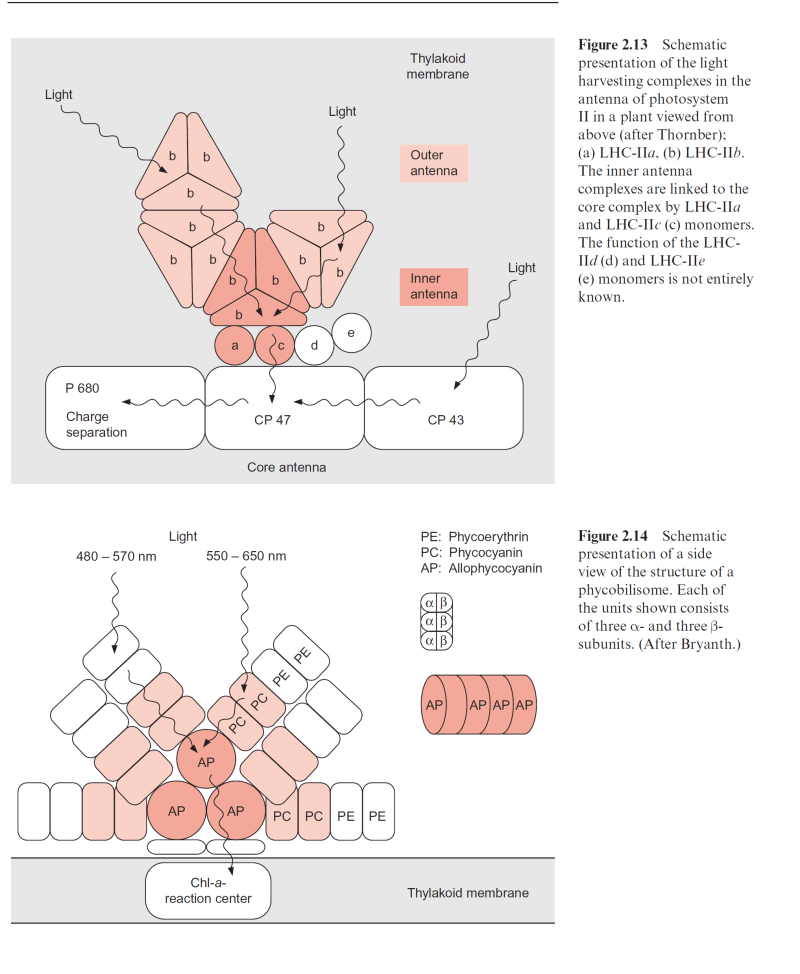
Figure 2.13 Schematic presentation of the light harvesting complexes in the antenna of photosystem II in a plant viewed from above (after Thornber); (a) LHC-IIa, (b) LHC-IIb. The inner antenna complexes are linked to the core complex by LHC-IIa and LHC-IIc (c) monomers. The function of the LHC- IId (d) and LHC-IIe (e) monomers is not entirely known
Figure 2.14 Schematic presentation of a side view of the structure of a phycobilisome. Each of the units shown consists of three - and three - subunits. (After Bryanth.)
Light-Harvesting: The Antenna Complex
phycobilisome : Energy and Electron Transfer in Photosynthetic Organisms,
Photosynthesis: What are Photosystems? Resonance, Reaction Centers and Antenna Complex?
Fate of light energy absorbed by the photosynthetic pigments, Fluorescence , phosphorescence,
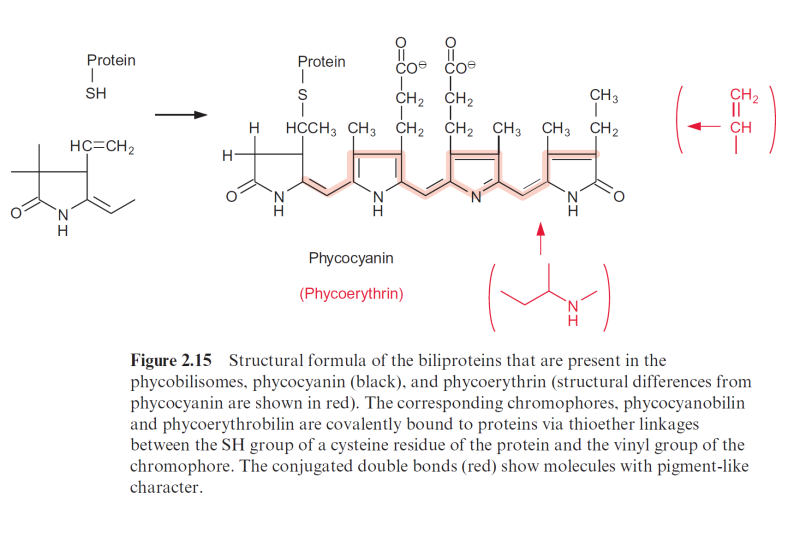
Figure 2.15 Structural formula of the biliproteins that are present in the phycobilisomes, phycocyanin (black), and phycoerythrin (structural differences from phycocyanin are shown in red). The corresponding chromophores, phycocyanobilin and phycoerythrobilin are covalently bound to proteins via thioether linkages between the SH group of a cysteine residue of the protein and the vinyl group of the chromophore. The conjugated double bonds (red) show molecules with pigment-like character.

