ESSENTIAL NUTRIENTS, DEFICIENCIES, AND PLANT DISORDERS
Only certain elements have been determined to be essential for plant growth. An essential element is defined as one whose absence prevents a plant from completing its life cycle (Arnon and Stout 1939) or one that has a clear physiological role (Epstein 1999). If plants are given these essential elements, as well as energy from sunlight, they can synthesize all the compounds they need for normal growth. Table 5.1 lists the elements that are considered to be essential for most, if not all, higher plants. The first three elements—hydrogen, carbon, and oxygen—are not considered mineral nutrients because they are obtained primarily from water or carbon dioxide.
Essential mineral elements are usually classified as macronutrients or micronutrients, according to their relative concentration in plant tissue. In some cases, the differences in tissue content of macronutrients and micronutrients are not as great as those indicated in Table 5.1. For example, some plant tissues, such as the leaf mesophyll, have almost as much iron or manganese as they do sulfur or magnesium. Many elements often are present in concentrations greater than the plant’s minimum requirements.
Some researchers have argued that a classification into macronutrients and micronutrients is difficult to justify physiologically. Mengel and Kirkby (1987) have proposed that the essential elements be classified instead according to their biochemical role and physiological function. Table 5.2 shows such a classification, in which plant nutrients have been divided into four basic groups:
- The first group of essential elements forms the organic (carbon carbon) compounds of the plant. Plants assimilate these nutrients via biochemical reactions involving oxidation and reduction.
- The second group is important in energy storage reactions or in maintaining structural integrity. Elements in this group are often present in plant tissues as phosphate, borate, and silicate esters in which the elemental group is bound to the hydroxyl group of an organic molecule (i.e., sugar–phosphate).
- The third group is present in plant tissue as either free ions or ions bound to substances such as the pectic acids present in the plant cell wall. Of particular importance are their roles as enzyme cofactors and in the regulation of osmotic potentials.
- The fourth group has important roles in reactions involving electron transfer.
Naturally occurring elements, other than those listed in Table 5.1, can also accumulate in plant tissues. For example, aluminum is not considered to be an essential element, but plants commonly contain from 0.1 to 500 ppm aluminum, and addition of low levels of aluminum to a nutrient solution may stimulate plant growth Many species in the genera Astragalus, Xylorhiza, and Stanleya accumulate selenium, although plants have not been shown to have a specific requirement for this element.
Cobalt is part of cobalamin (vitamin B12 and its derivatives), a component of several enzymes in nitrogen-fixing microorganisms. Thus cobalt deficiency blocks the development and function of nitrogen-fixing nodules. Nonetheless, plants that do not fix nitrogen, as well as nitrogen-fixing plants that are supplied with ammonium or nitrate, do not require cobalt. Crop plants normally contain only relatively small amounts of nonessential elements.
Special Techniques Are Used in Nutritional Studies
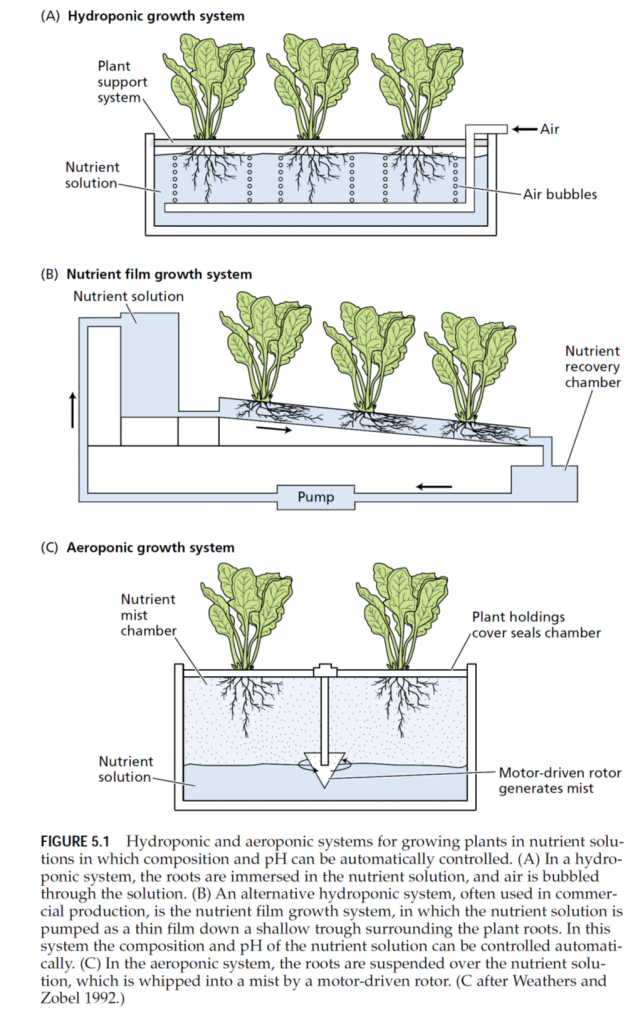
To demonstrate that an element is essential requires that plants be grown under experimental conditions in which only the element under investigation is absent. Such conditions are extremely difficult to achieve with plants grown in a complex medium such as soil. In the nineteenth century, several researchers, including Nicolas-Théodore de Saussure, Julius von Sachs, Jean-Baptiste-Joseph-Dieudonné Boussingault, and Wilhelm Knop, approached this problem by growing plants with their roots immersed in a nutrient solution containing only inorganic salts. Their demonstration that plants could grow normally with no soil or organic matter proved unequivocally that plants can fulfill all their needs from only inorganic elements and sunlight. The technique of growing plants with their roots immersed in nutrient solution without soil is called solution culture or hydroponics (Gericke 1937). Successful hydroponic culture (Figure 5.1A) requires a large volume of nutrient solution or frequent adjustment of the nutrient solution to prevent nutrient uptake by roots from producing radical changes in nutrient concentrations and pH of the medium. Asufficient supply of oxygen to the root system—also critical—may be achieved by vigorous bubbling of air through the medium. Hydroponics is used in the commercial production of many greenhouse crops. In one form of commercial hydroponic culture, plants are grown in a supporting material such as sand, gravel, vermiculite, or expanded clay (i.e., kitty litter). Nutrient solutions are then flushed through the supporting material, and old solutions are removed by leaching. In another form of hydroponic culture, plant roots lie on the surface of a trough, and nutrient solutions flow in a thin layer along the trough over the roots (Cooper 1979, Asher and Edwards 1983). This nutrient film growth system ensures that the roots receive an ample supply of oxygen (Figure 5.1B). Another alternative, which has sometimes been heralded as the medium of the future, is to grow the plants aeroponically (Weathers and Zobel 1992). In this technique, plants are grown with their roots suspended in air while being sprayed continuously with a nutrient solution (Figure 5.1C). This approach provides easy manipulation of the gaseous environment around the root, but it requires higher levels of nutrients than hydroponic culture does to sustain rapid plant growth. For this reason and other technical difficulties, the use of aeroponics is not widespread.
| TABLE 5.1 Adequate tissue levels of elements that may be required by plants | |||
| Chemical Element symbol | Concentration in dry matter (% or ppm) a | Relative number of atoms with respect to molybdenum | |
| Obtained from water or carbon dioxide | |||
| Hydrogen | H | 6 | 60,000,000 |
| Carbon | C | 45 | 40,000,000 |
| Oxygen | O | 45 | 30,000,000 |
| Obtained from the soil | |||
| Macronutrients | |||
| Nitrogen | N | 1.5 | 1,000,000 |
| Potassium | K | 1.0 | 250,000 |
| Calcium | Ca | 0.5 | 125,000 |
| Magnesium | Mg | 0.2 | 80,000 |
| Phosphorus | P | 0.2 | 60,000 |
| Sulfur | S | 0.1 | 30,000 |
| Silicon | Si | 0.1 | 30,000 |
| Micronutrients | |||
| Chlorine | Cl | 100 | 3,000 |
| Iron | Fe | 100 | 2,000 |
| Boron | B | 20 | 2,000 |
| Manganese | Mn | 50 | 1,000 |
| Sodium | Na | 10 | 400 |
| Zinc | Zn | 20 | 300 |
| Copper | Cu | 6 | 100 |
| Nickel | Ni | 0.1 | 2 |
| Molybdenum | Mo | 0.1 | 1 |
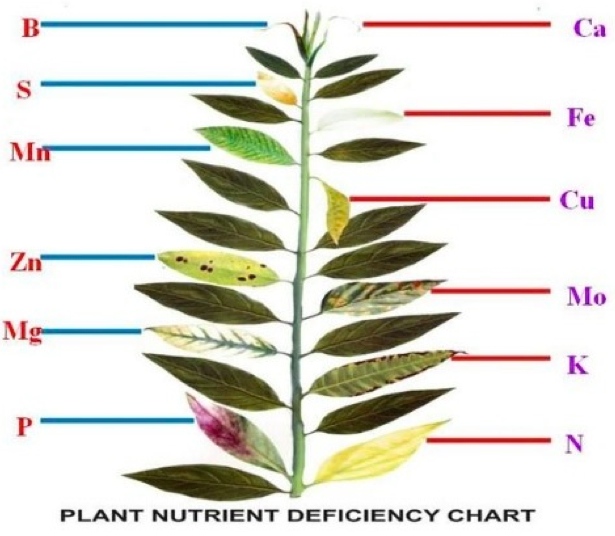
Identifying nutrient deficiency symptoms in field crops
Nutrient Solutions Can Sustain Rapid Plant Growth Over
the years, many formulations have been used for nutrient solutions. Early formulations developed by Knop in Germany included only KNO3, Ca(NO3)2, KH2PO4, MgSO4, and an iron salt. At the time this nutrient solution was believed to contain all the minerals required by the plant, but these experiments were carried out with chemicals that were contaminated with other elements that are now known to be essential (such as boron or molybdenum). Table 5.3 shows a more modern formulation for a nutrient solution. This formulation is called a modified Hoagland solution, named after Dennis R. Hoagland, a researcher who was prominent in the development of modern mineral nutrition research in the United States
A modified Hoagland solution contains all of the known mineral elements needed for rapid plant growth. The concentrations of these elements are set at the highest possible levels without producing toxicity symptoms or salinity stress and thus may be several orders of magnitude higher than those found in the soil around plant roots. For example, whereas phosphorus is present in the soil solution at concentrations normally less than 0.06 ppm, here it is offered at 62 ppm (Epstein 1972). Such high initial levels permit plants to be grown in a medium for extended periods without replenishment of the nutrients. Many researchers, however, dilute their nutrient solutions severalfold and replenish them frequently to minimize fluctuations of nutrient concentration in the medium and in plant tissue.
Another important property of the modified Hoagland formulation is that nitrogen is supplied as both ammonium (NH4 +) and nitrate (NO3 –). Supplying nitrogen in a balanced mixture of cations and anions tends to reduce the rapid rise in the pH of the medium that is commonly observed when the nitrogen is supplied solely as nitrate anion (Asher and Edwards 1983). Even when the pH of the medium is kept neutral, most plants grow better if they have access to both NH4 + and NO3 – because absorption and assimilation of the two nitrogen forms promotes cation–anion balance within the plant (Raven and Smith 1976; Bloom 1994).
A significant problem with nutrient solutions is maintaining the availability of iron. When supplied as an inorganic salt such as FeSO4 or Fe(NO3)2, iron can precipitate out of solution as iron hydroxide. If phosphate salts are present, insoluble iron phosphate will also form. Precipitation of the iron out of solution makes it physically unavailable to the plant, unless iron salts are added at frequent intervals. Earlier researchers approached this problem by adding iron together with citric acid or tartaric acid. Compounds such as these are called chelators because they form soluble complexes with cations such as iron and calcium in which the cation is held by ionic forces, rather than by covalent bonds. Chelated cations thus are physically more available to a plant. More modern nutrient solutions use the chemicals ethylenediaminetetraacetic acid (EDTA) or diethylenetriamine pentaacetic acid (DTPA, or pentetic acid) as chelating agents (Sievers and Bailar 1962). Figure 5.2 shows the structure of DTPA. The fate of the chelation complex during iron uptake by the root cells is not clear; iron may be released from the chelator when it is reduced from Fe3+ to Fe2+ at the root surface. The chelator may then diffuse back into the nutrient (or soil) solution and react with another Fe3+ ion or other metal ions. After uptake, iron is kept soluble by chelation with organic compounds present in plant cells. Citric acid may play a major role in iron chelation and its long-distance transport in the xylem.
Inadequate supply of an essential element results in a nutritional disorder manifested by characteristic deficiency symptoms. In hydroponic culture, withholding of an essential element can be readily correlated with a given set of symptoms for acute deficiencies. Diagnosis of soil-grown plants can be more complex, for the following reasons
- Both chronic and acute deficiencies of several elements may occur simultaneously.
- Deficiencies or excessive amounts of one element may induce deficiencies or excessive accumulations of another.
- Some virus-induced plant diseases may producesymptoms similar to those of nutrient deficiencies.
Nutrient deficiency symptoms in a plant are the expression of metabolic disorders resulting from the insufficient supply of an essential element. These disorders are related to the roles played by essential elements in normal plant metabolism and function. Table 5.2 lists some of the roles of essential elements.
Even though each essential element participates in many different metabolic reactions, some general statements about the functions of essential elements in plant metabolism are possible. In general, the essential elements function in plant structure, metabolic function, and osmoregulation of plant cells. More specific roles may be related to the ability of divalent cations such as calcium or magnesium to modify the permeability of plant membranes. In addition, research continues to reveal specific roles of these elements in plant metabolism; for example, calcium acts as a signal to regulate key enzymes in the cytosol (Hepler and Wayne 1985; Sanders et al. 1999). Thus, most essential elements have multiple roles in plant metabolism.
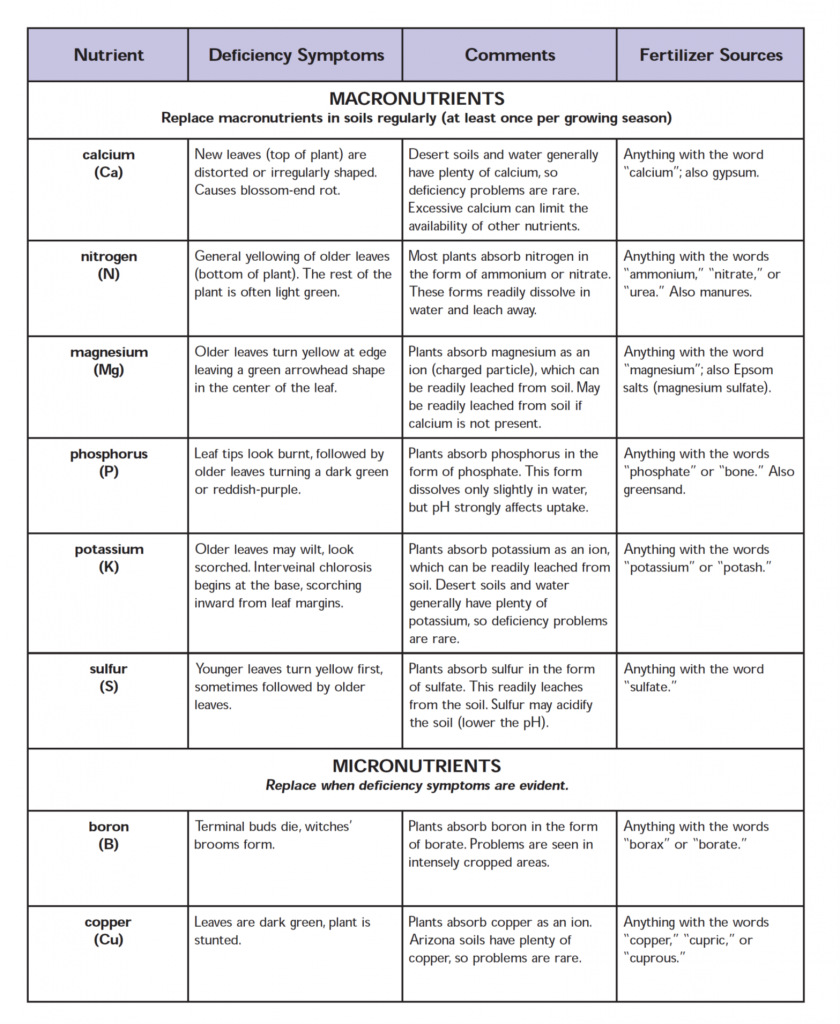
When relating acute deficiency symptoms to a particular essential element, an important clue is the extent to which an element can be recycled from older to younger leaves. Some elements, such as nitrogen, phosphorus, and potassium, can readily move from leaf to leaf; others, such as boron, iron, and calcium, are relatively immobile in most plant species (Table 5.4). If an essential element is mobile, deficiency symptoms tend to appear first in older leaves. Deficiency of an immobile essential element will become evident first in younger leaves. Although the precise mechanisms of nutrient mobilization are not well understood, plant hormones such as cytokinins appear to be involved (see Chapter 21). In the discussion that follows, we will describe the specific deficiency symptoms and functional roles for the mineral essential elements as they are grouped in Table 5.2.


Group 1: Deficiencies in mineral nutrients that are part of carbon compounds.
This first group consists of nitro- gen and sulfur. Nitrogen availability in soils limits plant productivity in most natural and agricultural ecosystems. By contrast, soils generally contain sulfur in excess. Nonetheless, nitrogen and sulfur share the property that their oxidation–reduction states range widely (see Chapter 12). Some of the most energy-intensive reactions in life con- vert the highly oxidized, inorganic forms absorbed from the soil into the highly reduced forms found in organic compounds such as amino acids.
NITROGEN.
Nitrogen is the mineral element that plants require in greatest amounts. It serves as a constituent of many plant cell components, including amino acids and nucleic acids. Therefore, nitrogen deficiency rapidly inhibits plant growth. If such a deficiency persists, most species show chlorosis (yellowing of the leaves), especially in the older leaves near the base of the plant (for pictures of nitro- gen deficiency and the other mineral deficiencies described in this chapter, see Web Topic 5.1). Under severe nitrogen deficiency, these leaves become completely yellow (or tan) and fall off the plant. Younger leaves may not show these symptoms initially because nitrogen can be mobilized from older leaves. Thus a nitrogen-deficient plant may have light green upper leaves and yellow or tan lower leaves.
When nitrogen deficiency develops slowly, plants may have markedly slender and often woody stems. This wood- iness may be due to a buildup of excess carbohydrates that cannot be used in the synthesis of amino acids or other nitrogen compounds. Carbohydrates not used in nitrogen metabolism may also be used in anthocyanin synthesis, leading to accumulation of that pigment. This condition is revealed as a purple coloration in leaves, petioles, and stems of some nitrogen-deficient plants, such as tomato and certain varieties of corn.
SULFUR.
Sulfur is found in two amino acids and is a con- stituent of several coenzymes and vitamins essential for metabolism. Many of the symptoms of sulfur deficiency are similar to those of nitrogen deficiency, including chlorosis, stunting of growth, and anthocyanin accumulation. This similarity is not surprising, since sulfur and nitrogen are both constituents of proteins. However, the chlorosis caused by sulfur deficiency generally arises initially in mature and young leaves, rather than in the old leaves as in nitrogen deficiency, because unlike nitrogen, sulfur is not easily remobilized to the younger leaves in most species. Nonetheless, in many plant species sulfur chlorosis may occur simultaneously in all leaves or even initially in the older leaves.
Group 2: Deficiencies in mineral nutrients that are impor- tant in energy storage or structural integrity.
This group consists of phosphorus, silicon, and boron. Phosphorus and silicon are found at concentrations within plant tissue that warrant their classification as macronutrients, whereas boron is much less abundant and considered a micronutri- ent. These elements are usually present in plants as ester linkages to a carbon molecule.
Phosphorus (as phosphate, PO 3–) is an inte- gral component of important compounds of plant cells, including the sugar–phosphate intermediates of respiration and photosynthesis, and the phospholipids that make up plant membranes. It is also a component of nucleotides used in plant energy metabolism (such as ATP) and in DNA and RNA. Characteristic symptoms of phosphorus deficiency include stunted growth in young plants and a dark green coloration of the leaves, which may be mal- formed and contain small spots of dead tissue called necrotic spots
As in nitrogen deficiency, some species may produce excess anthocyanins, giving the leaves a slight purple col- oration. In contrast to nitrogen deficiency, the purple col- oration of phosphorus deficiency is not associated with chlorosis. In fact, the leaves may be a dark greenish purple. Additional symptoms of phosphorus deficiency include the production of slender (but not woody) stems and the death of older leaves. Maturation of the plant may also be delayed.
SILICON.
Only members of the family Equisetaceae—called scouring rushes because at one time their ash, rich in gritty silica, was used to scour pots—require silicon to complete their life cycle. Nonetheless, many other species accumu- late substantial amounts of silicon within their tissues and show enhanced growth and fertility when supplied with adequate amounts of silicon (Epstein 1999).
Plants deficient in silicon are more susceptible to lodg- ing (falling over) and fungal infection. Silicon is deposited primarily in the endoplasmic reticulum, cell walls, and intercellular spaces as hydrated, amorphous silica (SiO2·nH2O). It also forms complexes with polyphenols and thus serves as an alternative to lignin in the reinforcement of cell walls. In addition, silicon can ameliorate the toxicity of many heavy metals.
BORON.
Although the precise function of boron in plant metabolism is unclear, evidence suggests that it plays roles in cell elongation, nucleic acid synthesis, hormone responses, and membrane function (Shelp 1993). Boron- deficient plants may exhibit a wide variety of symptoms, depending on the species and the age of the plant.
A characteristic symptom is black necrosis of the young leaves and terminal buds. The necrosis of the young leaves occurs primarily at the base of the leaf blade. Stems may be unusually stiff and brittle. Apical dominance may also be lost, causing the plant to become highly branched; how- ever, the terminal apices of the branches soon become necrotic because of inhibition of cell division. Structures such as the fruit, fleshy roots, and tubers may exhibit necro- sis or abnormalities related to the breakdown of internal tissues.
Group 3: Deficiencies in mineral nutrients that remain in ionic form.
This group includes some of the most familiar mineral elements: The macronutrients potassium, calcium, and magnesium, and the micronutrients chlorine, manganese, and sodium. They may be found in solution in the cytosol or vacuoles, or they may be bound electrostati- cally or as ligands to larger carbon-containing compounds.
Potassium, present within plants as the cation K+, plays an important role in regulation of the osmotic potential of plant cells (see Chapters 3 and 6). It also acti- vates many enzymes involved in respiration and photo- synthesis. The first observable symptom of potassium defi- ciency is mottled or marginal chlorosis, which then develops into necrosis primarily at the leaf tips, at the mar- gins, and between veins. In many monocots, these necrotic lesions may initially form at the leaf tips and margins and then extend toward the leaf base.
Because potassium can be mobilized to the younger leaves, these symptoms appear initially on the more mature leaves toward the base of the plant. The leaves may also curl and crinkle. The stems of potassium-deficient plants may be slender and weak, with abnormally short internodal regions. In potassium-deficient corn, the roots may have an increased susceptibility to root-rotting fungi present in the soil, and this susceptibility, together with effects on the stem, results in an increased tendency for the plant to be easily bent to the ground (lodging).
CALCIUM.
Calcium ions (Ca2+) are used in the synthesis of new cell walls, particularly the middle lamellae that sepa- rate newly divided cells. Calcium is also used in the mitotic spindle during cell division. It is required for the normal functioning of plant membranes and has been implicated as a second messenger for various plant responses to both environmental and hormonal signals (Sanders et al. 1999). In its function as a second messenger, calcium may bind to calmodulin, a protein found in the cytosol of plant cells. The calmodulin–calcium complex regulates many meta- bolic processes.
Characteristic symptoms of calcium deficiency include necrosis of young meristematic regions, such as the tips of roots or young leaves, where cell division and wall forma- tion are most rapid. Necrosis in slowly growing plants may be preceded by a general chlorosis and downward hook- ing of the young leaves. Young leaves may also appear deformed. The root system of a calcium-deficient plant may appear brownish, short, and highly branched. Severe stunting may result if the meristematic regions of the plant die prematurely.
MAGNESIUM.
In plant cells, magnesium ions (Mg2+) have a specific role in the activation of enzymes involved in respi- ration, photosynthesis, and the synthesis of DNA and RNA. Magnesium is also a part of the ring structure of the chloro- phyll molecule (see Figure 7.6A). A characteristic symptom of magnesium deficiency is chlorosis between the leaf veins, occurring first in the older leaves because of the mobility of this element. This pattern of chlorosis results because the chlorophyll in the vascular bundles remains unaffected for longer periods than the chlorophyll in the cells between the bundles does. If the deficiency is extensive, the leaves may become yellow or white. An additional symptom of mag- nesium deficiency may be premature leaf abscission.
CHLORINE.
The element chlorine is found in plants as the chloride ion (Cl–). It is required for the water-splitting reac- tion of photosynthesis through which oxygen is produced (see Chapter 7) (Clarke and Eaton-Rye 2000). In addition, chlorine may be required for cell division in both leaves and roots (Harling et al. 1997). Plants deficient in chlorine develop wilting of the leaf tips followed by general leaf chlorosis and necrosis. The leaves may also exhibit reduced growth. Eventually, the leaves may take on a bronzelike color (“bronzing”). Roots of chlorine-deficient plants may appear stunted and thickened near the root tips.
Chloride ions are very soluble and generally available in soils because seawater is swept into the air by wind and is delivered to soil when it rains. Therefore, chlorine defi- ciency is unknown in plants grown in native or agricultural habitats. Most plants generally absorb chlorine at levels much higher than those required for normal functioning.
MANGANESE.
Manganese ions (Mn2+) activate several enzymes in plant cells. In particular, decarboxylases and dehydrogenases involved in the tricarboxylic acid (Krebs) cycle are specifically activated by manganese. The best- defined function of manganese is in the photosynthetic reaction through which oxygen is produced from water (Marschner 1995). The major symptom of manganese defi- ciency is intervenous chlorosis associated with the devel- opment of small necrotic spots. This chlorosis may occur on younger or older leaves, depending on plant species and growth rate.
SODIUM.
Most species utilizing the C4 and CAM pathways of carbon fixation (see Chapter 8) require sodium ions (Na+). In these plants, sodium appears vital for regenerat- ing phosphoenolpyruvate, the substrate for the first car-boxylation in the C4 and CAM pathways (Johnstone et al. 1988). Under sodium deficiency, these plants exhibit chloro- sis and necrosis, or even fail to form flowers. Many C3 species also benefit from exposure to low levels of sodium ions. Sodium stimulates growth through enhanced cell expansion, and it can partly substitute for potassium as an osmotically active solute.
Group 4: Deficiencies in mineral nutrients that are involved in redox reactions.
This group of five micronu- trients includes the metals iron, zinc, copper, nickel, and molybdenum. All of these can undergo reversible oxidations and reductions (e.g., Fe2+ ~ Fe3+) and have important roles in electron transfer and energy transformation. They are usu- ally found in association with larger molecules such as cytochromes, chlorophyll, and proteins (usually enzymes).
IRON.
Iron has an important role as a component of enzymes involved in the transfer of electrons (redox reac- tions), such as cytochromes. In this role, it is reversibly oxi- dized from Fe2+ to Fe3+ during electron transfer. As in mag- nesium deficiency, a characteristic symptom of iron deficiency is intervenous chlorosis. In contrast to magne- sium deficiency symptoms, these symptoms appear ini- tially on the younger leaves because iron cannot be readily mobilized from older leaves. Under conditions of extreme or prolonged deficiency, the veins may also become chlorotic, causing the whole leaf to turn white.
The leaves become chlorotic because iron is required for the synthesis of some of the chlorophyll–protein complexes in the chloroplast. The low mobility of iron is probably due to its precipitation in the older leaves as insoluble oxides or phosphates or to the formation of complexes with phyto- ferritin, an iron-binding protein found in the leaf and other plant parts (Oh et al. 1996). The precipitation of iron dimin- ishes subsequent mobilization of the metal into the phloem for long-distance translocation.
ZINC.
Many enzymes require zinc ions (Zn2+) for their activity, and zinc may be required for chlorophyll biosyn- thesis in some plants. Zinc deficiency is characterized by a reduction in internodal growth, and as a result plants dis- play a rosette habit of growth in which the leaves form a circular cluster radiating at or close to the ground. The leaves may also be small and distorted, with leaf margins having a puckered appearance. These symptoms may result from loss of the capacity to produce sufficient amounts of the auxin indoleacetic acid. In some species (corn, sorghum, beans), the older leaves may become inter- venously chlorotic and then develop white necrotic spots. This chlorosis may be an expression of a zinc requirement for chlorophyll biosynthesis.
COPPER.
Like iron, copper is associated with enzymes involved in redox reactions being reversibly oxidized from Cu+ to Cu2+. An example of such an enzyme is plasto- cyanin, which is involved in electron transfer during the light reactions of photosynthesis (Haehnel 1984). The ini- tial symptom of copper deficiency is the production of dark green leaves, which may contain necrotic spots. The necrotic spots appear first at the tips of the young leaves and then extend toward the leaf base along the margins. The leaves may also be twisted or malformed. Under extreme copper deficiency, leaves may abscise prematurely.
NICKEL.
Urease is the only known nickel-containing enzyme in higher plants, although nitrogen-fixing microor- ganisms require nickel for the enzyme that reprocesses some of the hydrogen gas generated during fixation (hydrogen uptake hydrogenase) (see Chapter 12). Nickel- deficient plants accumulate urea in their leaves and, con- sequently, show leaf tip necrosis. Plants grown in soil sel- dom, if ever, show signs of nickel deficiency because the amounts of nickel required are minuscule.
MOLYBDENUM.
Molybdenum ions (Mo4+ through Mo6+) are components of several enzymes, including nitrate reductase and nitrogenase. Nitrate reductase catalyzes the reduction of nitrate to nitrite during its assimilation by the plant cell; nitrogenase converts nitrogen gas to ammonia in nitrogen-fixing microorganisms (see Chapter 12). The first indication of a molybdenum deficiency is general chloro- sis between veins and necrosis of the older leaves. In some plants, such as cauliflower or broccoli, the leaves may not become necrotic but instead may appear twisted and sub- sequently die (whiptail disease). Flower formation may be prevented, or the flowers may abscise prematurely.
Because molybdenum is involved with both nitrate assimilation and nitrogen fixation, a molybdenum defi- ciency may bring about a nitrogen deficiency if the nitrogen source is primarily nitrate or if the plant depends on sym- biotic nitrogen fixation. Although plants require only small amounts of molybdenum, some soils supply inadequate levels. Small additions of molybdenum to such soils can greatly enhance crop or forage growth at negligible cost.
| TABLE 5.3 Composition of a modified Hoagland nutrient solution for growing plants | |||||||
| Compound | Molecular weight | Concentration of stock solution | Concentration of stock solution | Volume of stock solution per liter of final solution | Element | Final concentration of element | |
| g mol–1 | mM | g L–1 | mL | mM | ppm | ||
| Macronutrients | |||||||
| KNO3 Potassium nitrate | 101.10 | 1,000 | 101.10 | 6.0 | N | 16,000 | 224 |
| Ca(NO3)2×4H2O Calcium Nitrate Tetrahydrate | 236.16 | 1,000 | 236.16 | 4.0 | K | 6,000 | 235 |
| NH4H2PO4 Ammonium dihydrogen phosphate | 115.08 | 1,000 | 115.08 | 2.0 | Ca | 4,000 | 160 |
| MgSO4×7H2O | 246.48 | 1,000 | 246.49 | 1.0 | P | 2,000 | 62 |
| Heptahydrate, 2O (“Epsom salt”) Epsonite of Magnesium sulfate | S | 1,000 | 32 | ||||
| Mg | 1,000 | 24 | |||||
| Micronutrients | |||||||
| KCl | 74.55 | 25 | 1.864 | Cl | 50 | 1.77 | |
| H3BO3 Boric Acid | 61.83 | 12.5 | 0.773 | B | 25 | 0.27 | |
| MnSO4×H2O Manganese sulfate monohydrate | 169.01 | 1.0 | 0.169 | 2.0 | Mn | 2.0 | 0.11 |
| ZnSO4×7H2O Zinc sulfate heptahydrate | 287.54 | 1.0 | 0.288 | Zn | 2.0 | 0.13 | |
| CuSO4×5H2O Copper sulfate pentahydrate | 249.68 | 0.25 | 0.062 | Cu | 0.5 | 0.03 | |
| 161.97 | 0.25 | 0.040 | Mo | 0.5 | 0.05 | ||
| NaFe DTPA (10% Fe) | 468.20 | 64 | 30.0 | 0.3–1.0 | Fe | 16.1–53.7 1.00–3.00 | |
| Optionala | |||||||
| NiSO4×6H2O Nickel sulfate hexahydrate | 262.86 | 0.25 | 0.066 | 2.0 | Ni | 0.5 | 0.03 |
| Na2SiO3×9H2O Sodium metasilicate nonahydrate | 284.20 | 1,000 | 284.20 | 1.0 | Si | 1,000 | 28 |

| Chelates Several factors reduce the bioavailability of Fe, including high soil pH, high bicarbonate content, plant species (grass species are usually more efficient than other species because they can excrete effective ligands), and abiotic stresses. Plants typically utilize iron as ferrous iron (Fe2+). Ferrous iron can be readily oxidized to the plant-unavailable ferric form (Fe3+) when soil pH is greater than 5.3 (Morgan and Lahav 2007). Iron deficiency often occurs if soil pH is greater than 7.4. Chelated iron can prevent this conversion from Fe2+ to Fe3+. Applying nutrients such as Fe, Mn, Zn, and Cu directly to the soil is inefficient because in soil solution they are present as positively charged metal ions and will readily react with oxygen and/or negatively charged hydroxide ions (OH-). If they react with oxygen or hydroxide ions, they form new compounds that are not bioavailable to plants. Both oxygen and hydroxide ions are abundant in soil and soilless growth media. The ligand can protect the micronutrient from oxidization or precipitation. Figure 1 shows examples of the typical iron deficiency symptoms of lychee grown in Homestead, Florida, in which the lychee trees have yellow leaves and small, abnormal fruits. Applying chelated fertilizers is an easy and practical correction method to avoid this nutrient disorder. For example, the oxidized form of iron is ferric (Fe3+), which is not bioavailable to plants and usually forms brown ferric hydroxide precipitation (Fe(OH)3). Ferrous sulfate, which is not a chelated fertilizer, is often used as the iron source. Its solution should be green. If the solution turns brown, the bioavailable form of iron has been oxidized and Fe is therefore unavailable to plants. Figure 3. Comparison of foliar applications of chelated Fe, regular iron fertilizers, and no iron fertilization for correcting iron deficiency of lychee (Litchi chinensis, the soapberry family). Credit: Yuncong Li, UF/IFAS [Pentetic acid or diethylenetriaminepentaacetic acid (DTPA)] The ligands EDTA, DTPA, and EDDHA are often used in chelated fertilizers (Table 4). Their effectiveness differs significantly. EDDHA chelated Fe is most stable at soil pH greater than 7 (Figure 4, A and B). Chelated fertilizer stability is desired because it means the chelated micronutrient will remain in a bioavailable form for a much longer time period, thus increasing micronutrient use efficiency in vegetable and fruit production. The stability of three typical chelated Fe fertilizers varies at different pH conditions (Figure 4, A). The Y-axis represents the ratio of chelated Fe to total chelate and ranges from 0 to 1.0. A value of 1.0 means the chelate is stable. The X-axis represents soil pH. At 6.0, the ratios for all three chelated Fe fertilizers are 1.0 (stable), but at pH 7.5, only the ratio of EDDTA chelated Fe is 1.0. That of DTPA chelated Fe is only 0.5, and that of EDTA chelated Fe is only 0.025. So, in practice, EDDTA chelated Fe fertilizer is most effective when pH is greater than 7 but most costly. Accordingly, crop yields of these three chelated fertilizers are in this order: FeEDDHA > FeDTPA > FeEDTA (Figure 4, B). See Micronutrient Deficiencies in Citrus: Iron, Zinc, and Manganese (https://edis.ifas.ufl.edu/ss423) for effective pH ranges of iron chelates. Table 3 shows the relationship between soil pH and chelated fertilizer requirement. Correction of Fe deficiency depends on individual crop response and many other factors. For instance, for vegetables, the rate is usually 0.4–1 lb. chelated Fe in 100 gal. of water per acre. Deciduous fruits need 0.1–0.2 lb. chelated Fe in 25 gal. of water per acre (Table 5). Foliar application is more effective than soil application. For foliar application, either inorganic or chelated Fe is effective, but for fertigation, chelated Fe should be used. In high pH soil, crops are also vulnerable to Cu deficiency stresses. Chelated Cu is significantly more effective than inorganic Cu. A commonly used copper chelate is Na2CuEDTA, which contains 13% Cu. Natural organic materials have approximately 0.5% Cu (Table 5). In addition to soil pH, Mn is also influenced by aeration, moisture, and organic matter content. Chelated Mn can improve Mn bioavailability. Mn deficiency occurs more often in high pH and dry soil. Similar to other micronutrients, foliar spray is much more effective than soil application. For commercial vegetable production, 0.2–0.5 lb. MnEDTA in 200 gal. of water per acre can effectively correct Mn deficiency (Table 5). Zinc is another micronutrient whose bioavailability is closely associated with soil pH. Crops may be susceptible to Zn deficiency in soil with pH > 7.3. Spraying 0.10–0.14 lb. chelated Zn in 100 gal. of water per acre is effective (Poh et al. 2009). Animal waste and municipal waste also contain Cu, Mn, and Zn micronutrients (Table 5). For more information about micronutrient deficiency in crops, see Plant Tissue Analysis and Interpretation for Vegetable Crops in Florida (https://edis.ifas.ufl.edu/ep081), |
| TABLE 5.5 Comparison of properties of three major types of silicate clays found in the soil | |||
| Property | Type of clay | ||
| Montmorillonite | Illite | Kaolinite | |
| Size (µm) | 0.01–1.0 | 0.1–2.0 | 0.1–5.0 |
| Shape | Irregular flakes | Irregular flakes | Hexagonal crystals |
| Cohesion | High | Medium | Low |
| Water-swelling capacity | High | Medium | Low |
| Cation exchange capacity (milliequivalents 100 g-1) | 80–100 | 15–40 | 3–15 |
| TABLE 6.1 Comparison of observed and predicted ion concentrations in pea root tissue | |||
| Ion | Concentration in external medium (mmol L–1) | Internal concentration (mmol L–1) | |
| Predicted | Observed | ||
| K+ Na+ Mg2+ Ca2+ NO – 3 Cl– H2PO – 4 SO 2– 4 | 1 1 0.25 1 2 1 1 0.25 | 74 74 1340 5360 0.0272 0.0136 0.0136 0.00005 | 75 8 3 2 28 7 21 19 |
| TABLE 6.2 The vacuolar pH of some hyperacidifying plant species | ||
| Tissue | Species | pHa |
| Fruits | Lime (Citrus aurantifolia) Lemon (Citrus limonia) Cherry (Prunuscerasus) Grapefruit (Citrus paradisi) Rosette oxalis (Oxalisdeppei) Wax begonia (Begonia semperflorens) Begonia ‘Lucerna’ Oxalis sp. Sorrel (Rumex sp.) Prickly Pear (Opuntia phaeacantha)b | |
| 1.7 | ||
| 2.5 | ||
| 2.5 | ||
| 3.0 | ||
| Leaves | ||
| 1.3 | ||
| 1.5 | ||
| 0.9 – 1.4 | ||
| 1.9 – 2.6 | ||
| 2.6 | ||
| 1.4 (6:45A.M.) | ||
| 5.5 (4:00 P.M.) |
.
Analysis of Plant Tissues Reveals Mineral Deficiencies
Requirements for mineral elements change during the growth and development of a plant. In crop plants, nutri- ent levels at certain stages of growth influence the yield of the economically important tissues (tuber, grain, and so on). To optimize yields, farmers use analyses of nutrient levels in soil and in plant tissue to determine fertilizer schedules.
Soil analysis is the chemical determination of the nutri- ent content in a soil sample from the root zone. As dis- cussed later in the chapter, both the chemistry and the biol- ogy of soils are complex, and the results of soil analyses vary with sampling methods, storage conditions for the samples, and nutrient extraction techniques. Perhaps more important is that a particular soil analysis reflects the lev- els of nutrients potentially available to the plant roots from the soil, but soil analysis does not tell us how much of a particular mineral nutrient the plant actually needs or is able to absorb. This additional information is best deter- mined by plant tissue analysis.
Proper use of plant tissue analysis requires an under- standing of the relationship between plant growth (or yield) and the mineral concentration of plant tissue sam- ples (Bouma 1983). As the data plot in Figure 5.3 shows, when the nutrient concentration in a tissue sample is low, growth is reduced. In this deficiency zone of the curve, an increase in nutrient availability is directly related to an increase in growth or yield. As the nutrient availability con- tinues to increase, a point is reached at which further addi- tion of nutrients is no longer related to increases in growth or yield but is reflected in increased tissue concentrations. This region of the curve is often called the adequate zone. The transition between the deficiency and adequate zones of the curve reveals the critical concentration of the nutrient (see Figure 5.3), which may be defined as the min- imum tissue content of the nutrient that is correlated with maximal growth or yield. As the nutrient concentration of the tissue increases beyond the adequate zone, growth or yield declines because of toxicity (this is the toxic zone).
To evaluate the relationship between growth and tissue nutrient concentration, researchers grow plants in soil or nutrient solution in which all the nutrients are present in adequate amounts except the nutrient under consideration. At the start of the experiment, the limiting nutrient is added in increasing concentrations to different sets of plants, and the concentrations of the nutrient in specific tis- sues are correlated with a particular measure of growth or yield. Several curves are established for each element, one for each tissue and tissue age.
Because agricultural soils are often limited in the ele- ments nitrogen, phosphorus, and potassium, many farm- ers routinely use, at a minimum, curves for these elements. If a nutrient deficiency is suspected, steps are taken to cor- rect the deficiency before it reduces growth or yield. Plant analysis has proven useful in establishing fertilizer sched- ules that sustain yields and ensure the food quality of many crops.
TREATING NUTRITIONAL DEFICIENCIES
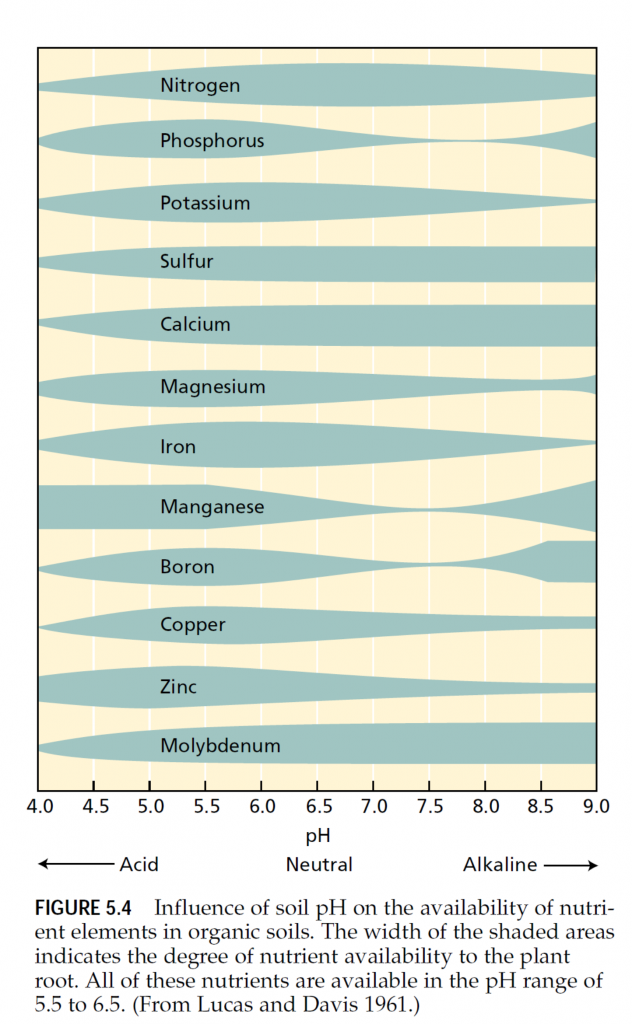
Many traditional and subsistence farming practices pro- mote the recycling of mineral elements. Crop plants absorb the nutrients from the soil, humans and animals consume locally grown crops, and crop residues and manure from humans and animals return the nutrients to the soil. The main losses of nutrients from such agricultural systems ensue from leaching that carries dissolved ions away with drainage water. In acid soils, leaching may be decreased by the addition of lime—a mix of CaO, CaCO3, and Ca(OH)2—to make the soil more alkaline because many mineral elements form less soluble compounds when the pH is higher than 6 (Figure 5.4).
In the high-production agricultural systems of industrial countries, the unidirectional removal of nutrients from the soil to the crop can become significant because a large por- tion of crop biomass leaves the area of cultivation. Plants synthesize all their components from basic inorganic sub- stances and sunlight, so it is important to restore these lost nutrients to the soil through the addition of fertilizers.

Crop Yields Can Be Improved by Addition of Fertilizers
Most chemical fertilizers contain inorganic salts of the macronutrients nitrogen, phosphorus, and potassium (see Table 5.1). Fertilizers that contain only one of these three nutrients are termed straight fertilizers. Some examples of straight fertilizers are superphosphate, ammonium nitrate, and muriate of potash (a source of potassium). Fertilizers that contain two or more mineral nutrients are called com- pound fertilizers or mixed fertilizers, and the numbers on the package label, such as 10-14-10, refer to the effective per- centages of N, P2O5, and K2O, respectively, in the fertilizer.
With long-term agricultural production, consumption of micronutrients can reach a point at which they, too, must be added to the soil as fertilizers. Adding micronutrients to the soil may also be necessary to correct a preexisting defi- ciency. For example, some soils in the United States are
water and oxygen availability, and the type and number of microorganisms present in the soil.
As a consequence, the rate of mineralization is highly variable, and nutrients from organic residues become avail- able to plants over periods that range from days to months to years. The slow rate of mineralization hinders efficient fertilizer use, so farms that rely solely on organic fertilizers may require the addition of substantially more nitrogen or phosphorus and suffer even higher nutrient losses than farms that use chemical fertilizers. Residues from organic fertilizers do improve the physical structure of most soils, enhancing water retention during drought and increasing drainage in wet weather.
| TABLE 5.4 Mineral elements classified on the basis of their mobility within a plant and their tendency to retranslocate during deficiencies | |
| Mobile | Immobile |
| Nitrogen | Calcium |
| Potassium | Sulfur |
| Magnesium | Iron |
| Phosphorus | Boron |
| Chlorine | Copper |
| Sodium | |
| Zinc | |
| Molybdenum |
Some Mineral Nutrients Can Be Absorbed by Leaves
In addition to nutrients being added to the soil as fertiliz- ers, some mineral nutrients can be applied to the leaves as sprays, in a process known as foliar application, and the leaves can absorb the applied nutrients. In some cases, this method can have agronomic advantages over the applica- tion of nutrients to the soil. Foliar application can reduce the lag time between application and uptake by the plant, which could be important during a phase of rapid growth. It can also circumvent the problem of restricted uptake of a nutrient from the soil. For example, foliar application of mineral nutrients such as iron, manganese, and copper may be more efficient than application through the soil
deficient in boron, copper, zinc, manganese, molybdenum, or iron (Mengel and Kirkby 1987) and can benefit from nutrient supplementation.
Chemicals may also be applied to the soil to modify soil pH. As Figure 5.4 shows, soil pH affects the availability of all mineral nutrients. Addition of lime, as mentioned previ- ously, can raise the pH of acidic soils; addition of elemental sulfur can lower the pH of alkaline soils. In the latter case, microorganisms absorb the sulfur and subsequently release sulfate and hydrogen ions that acidify the soil.
Organic fertilizers, in contrast to chemical fertilizers, originate from the residues of plant or animal life or from natural rock deposits. Plant and animal residues contain many of the nutrient elements in the form of organic com- pounds. Before crop plants can acquire the nutrient ele- ments from these residues, the organic compounds must be broken down, usually by the action of soil microorgan- isms through a process called mineralization. Mineraliza- tion depends on many factors, including temperature,
available to the root system.
Nutrient uptake by plant leaves is most effective when the nutrient solution remains on the leaf as a thin film (Mengel and Kirkby 1987). Production of a thin film often requires that the nutrient solutions be supplemented with surfactant chemicals, such as the detergent Tween 80, that reduce surface tension. Nutrient movement into the plant seems to involve diffusion through the cuticle and uptake by leaf cells. Although uptake through the stomatal pore could provide a pathway into the leaf, the architecture of the pore (see Figures 4.13 and 4.14) largely prevents liquid penetration (Ziegler 1987).
For foliar nutrient application to be successful, damage to the leaves must be minimized. If foliar sprays are applied on a hot day, when evaporation is high, salts may accumulate on the leaf surface and cause burning or scorching. Spraying on cool days or in the evening helps to alleviate this problem. Addition of lime to the spray dimin- ishes the solubility of many nutrients and limits toxicity. Foliar application has proved economically successful mainly with tree crops and vines such as grapes, but it is also used with cereals. Nutrients applied to the leaves could save an orchard or vineyard when soil-applied nutri- ents would be too slow to correct a deficiency. In wheat, nitrogen applied to the leaves during the later stages of growth enhances the protein content of seeds.
…

SUMMARY
Plants are autotrophic organisms capable of using the energy from sunlight to synthesize all their components from carbon dioxide, water, and mineral elements. Studies of plant nutrition have shown that specific mineral ele- ments are essential for plant life. These elements are clas- sified as macronutrients or micronutrients, depending on the relative amounts found in plant tissue.
Certain visual symptoms are diagnostic for deficiencies in specific nutrients in higher plants. Nutritional disorders occur because nutrients have key roles in plant metabolism. They serve as components of organic compounds, in energy storage, in plant structures, as enzyme cofactors, and in electron transfer reactions. Mineral nutrition can be studied through the use of hydroponics or aeroponics, which allow the characterization of specific nutrient requirements. Soil and plant tissue analysis can provide information on the nutritional status of the plant–soil sys- tem and can suggest corrective actions to avoid deficien- cies or toxicities.
When crop plants are grown under modern high-pro- duction conditions, substantial amounts of nutrients are removed from the soil. To prevent the development of defi- ciencies, nutrients can be added back to the soil in the form of fertilizers. Fertilizers that provide nutrients in inorganic forms are called chemical fertilizers; those that derive from plant or animal residues are considered organic fertilizers. In both cases, plants absorb the nutrients primarily as inor- ganic ions. Most fertilizers are applied to the soil, but some are sprayed on leaves.
…
