CEC and Soil Nutrient Availability
Soil pH
Soil pH defines the relative acidity or alkalinity of the soil solution (Table 1.). The pH scale in natural systems ranges from 0 to 14. A pH value of 7.0 is neutral. Values below 7.0 are acid and those above 7.0 are alkaline, or basic. Many agricultural soils have a soil pH between 5.5 and 6.5.
Soil pH is a measurement of hydrogen ion (H+) activity, or effective concentration, in a soil and water solution. Soil pH is expressed in logarithmic terms, which means that each unit change in soil pH amounts to a tenfold change in acidity or alkalinity. For example, a soil with a pH of 6.0 has 10 times as much active H+ as one with a pH of 7.0.
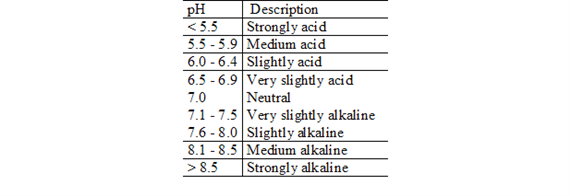
Descriptive terms commonly associated with certain ranges in soil pH are:
Cation and anion exchange capacity (CEC)
Cation-exchange capacity is defined as the degree to which a soil can adsorb and exchange cations.on surface with negative charge.

Sources of negative charge:

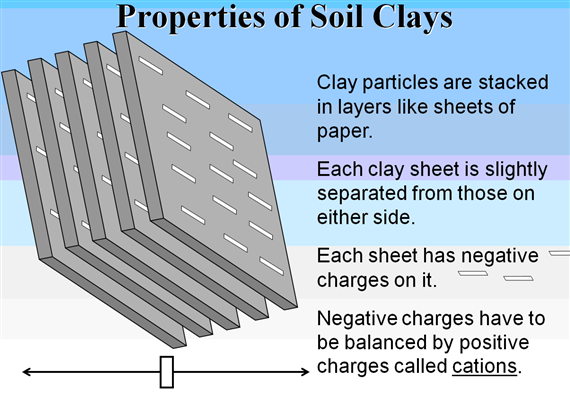

The main source of charge on clay minerals is isomorphous substitution which confers permanent charge on the surface of most layer silicates.
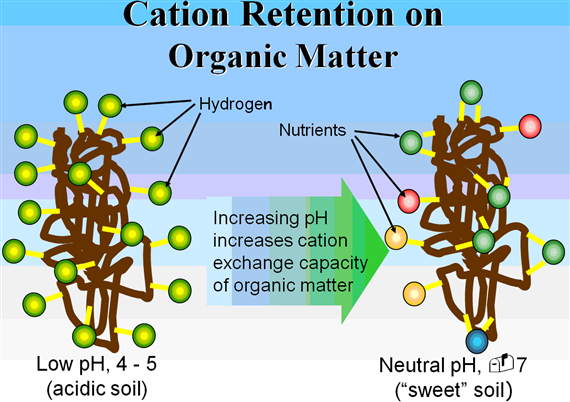
Ionization of hydroxyl groups on the surface of other soil colloids and organic matter can result in what is describes as pH dependent charges-mainly due to the dependent on the pH of the soil environment. Unlike permanent charges developed by isomorphous substitution, pH-dependent charges are variable and increase with increasing pH.

Presence of surface and broken – edge -OH groups gives the kaolinite clay particles their electronegativity and their capacity to absorb cations. In most soils there is a combination of constant and variable charge. Cation-a positively charged ion There are two types of cations, acidic or acid-forming cations, and basic, or alkaline-forming cations. The Hydrogen cation H+ and the Aluminum cation Al+++ are acid-forming.

The positively charged nutrients that we are mainly concerned with here are Calcium, Magnesium, Potassium and Sodium. These are all alkaline cations, also called basic cations or bases. Both types of cations may be adsorbed onto either a clay particle or soil organic matter (SOM). All of the nutrients in the soil need to be held there somehow, or they will just wash away when you water the garden or get a good rainstorm. Clay particles almost always have a negative (-) charge, so they attract and hold positively (+) charged nutrients and non-nutrients. Soil organic matter (SOM) has both positive and negative charges, so it can hold on to both cations and anions.( http://www.soilminerals.com/Cation_Exchange_Simplified.htm)
Anion-a negatively charged ion (NO3–, PO42-, SO42-, etc…)
Soil particles and organic matter have negative charges on their surfaces. Mineral cations can adsorb to the negative surface charges or the inorganic and organic soil particles. Once adsorbed, these minerals are not easily lost when the soil is leached by water and they also provide a nutrient reserve available to plant roots.
These minerals can then be replaced or exchanged by other cations (i.e., cation exchange)
vThe exchage processes (Figure 23) are REVERSIBLE (unless something precipitates, volatilizes, or is strongly adsorbed).

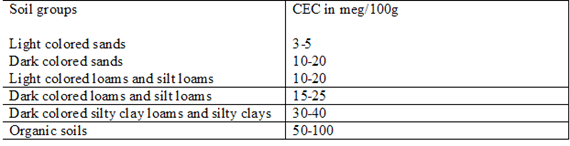
CEC is highly dependent upon soil texture and organic matter content Table 3, 4.). In general, the more clay and organic matter in the soil, the higher the CEC. Clay content is important because these small particles have a high ration of surface area to volume. Different types of clays also vary in CEC. Smectites have the highest CEC (80-100 millequivalents 100 g-1), followed by illites (15-40 meq 100 g-1) and kaolinites (3-15 meq 100 g-1).
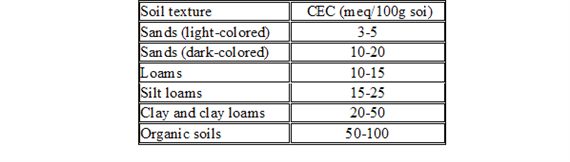
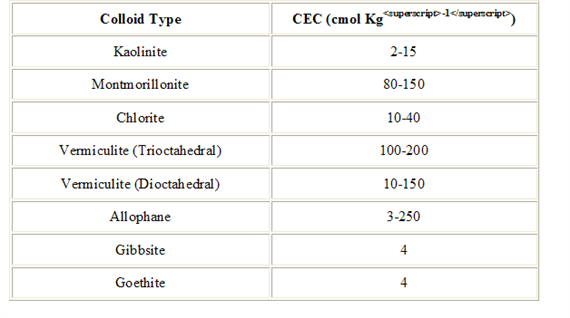
Measurement of CEC.
The CEC of soil is usually measured by saturating the soil with an index cation such as Na+, removal of the excess salts of the index cation with a dilute solution, and then displacing the Na+ with another cation. The amount of Na+ displaced is then measured and the CEC is calculated.
In general, the CEC of most soils increases with an increase in soil pH. Two factors determine the relative proportions of the different cations adsorbed by clays. First, cations are not held equally tight by the soil colloids. When the cations are present in equivalent amounts, the order of strength of adsorption is Al3+ > Ca2+ > Mg2+ > K+ = NH4+ > Na+.
The relative concentrations of the cations in soil solution helps determine the degree of adsorption. Very acid soils will have high concentrations of H+ and Al3+. In neutral to moderately alkaline soils, Ca2+ and Mg2+ dominate. Poorly drained arid soils may adsorb Na in very high quantities.

Base saturation
The proportion of CEC satisfied by basic cations (Ca, Mg, K, and Na) is termed percentage base saturation (BS%). This property is inversely related to soil acidity. As the BS% increases, the pH increases. High base saturation is preferred but not essential for tree fruit production. The availability of nutrient cations such as Ca, Mg, and K to plants increases with increasing BS%.
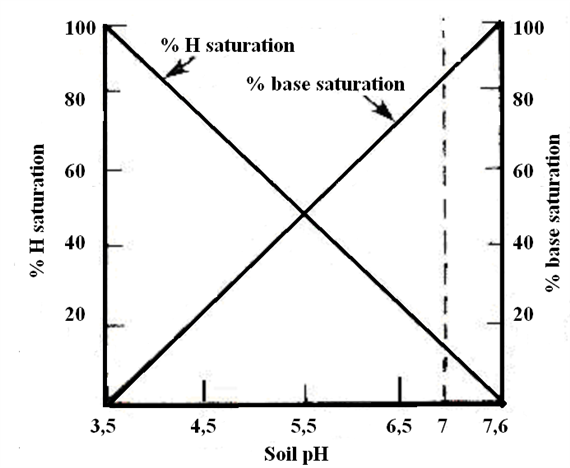
Base saturation is usually close to 100% in arid region soils. Base saturation below 100% indicates that part of the CEC is occupied by hydrogen and/or aluminum ions. Base saturation above 100% indicates that soluble salts or lime may be present, or that there is a procedural problem with the analysis.
CEC and availability of nutrients
Exchangeable cations, may become available to plants. Plant roots also possess cation exchange capacity. Hydrogen ions from the root hairs and microorganisms may replace nutrient cations from the exchange complex on soil colloids. The nutrient cations are then released into the soil solution where they can be taken up by the adsorptive surfaces of roots and soil organisms. They may however, be lost from the system by drainage water.

Additionally, high levels of one nutrient may influence uptake of another (antagonistic relationship). For example, K uptake by plants is limited by high levels of Ca in some soils. High levels of K can in turn, limit Mg uptake even if Mg levels in soil are high.
Anion-exchange capacity (AEC)
Sources of anion exchange capacity
Anion exchange arise from the protonation of hydroxyl groups on the edges of silicate clays and on the surfaces of metal oxide clays Anion exchange is inversely related with pH is greatest in soils dominated by the sesquioxides. The anions Cl–, NO3–, and SeO42- and to some extent HS– ands SO42-, HCO3–, and CO3– adsorb mainly by ion exchange. Borate, phospahate and carboxylate adsorb principally by specific adsorption mechanisms. (http://jan.ucc.nau.edu/~doetqp-p/courses/env320/lec13/Lec13.html)
The total exchangeable anions that a soil can adsorb, measured as milliequivalents per 100 grams of soil. (http://www.encyclopedia.com/doc/1O7-anionexchangecapacity.html )
In contrast to CEC, AEC is the degree to which a soil can adsorb and exchange anions. AEC increases as soil pH decreases. The pH of most productive soils is usually too high (exceptions are for volcanic soils) for full development of AEC and thus it generally plays a minor role in supplying plants with anions.

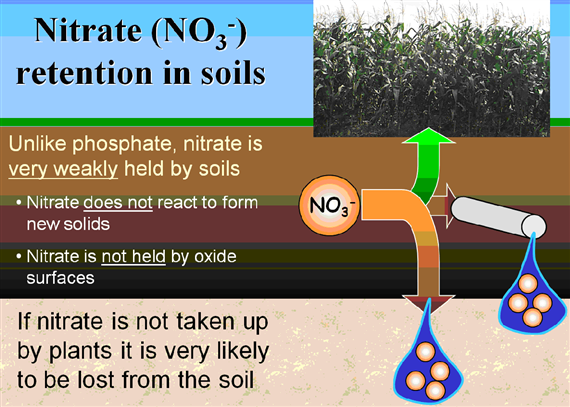
Because the AEC of most agricultural soils is small compared to their CEC, mineral anions such as nitrate (NO3– and Cl–) are repelled by the negative charge on soil colloids. These ions remain mobile in the soil solution and thus are susceptible to leaching. (http://soils.tfrec.wsu.edu/webnutritiongood/soilprops/04CEC.htm)
Phosphate anions are relativelly bounded on the positivelly charged places (iron, aluminium, calcium compounds etc.) (Figure 28).

Nitrate is weakly bounded and the nitrate compounds are well soluble soluble in water, that why nitrate can easily be leahed out (Figure 29).
Nitrification
NH4+ NO3– Some of the ammonium produced by decomposition is converted to nitrate via a process called nitrification. The bacteria that carry out this reaction gain energy from it. Nitrification requires the presence of oxygen, so nitrification can happen only in oxygen-rich environments like circulating or flowing waters and the very surface layers of soils and sediments. The process of nitrification has some important consequences. Ammonium ions are positively charged and therefore stick (are sorbed) to negatively charged clay particles and soil organic matter. The positive charge prevents ammonium nitrogen from being washed out of the soil (or leached) by rainfall. In contrast, the negatively charged nitrate ion is not held by soil particles and so can be washed down the soil profile, leading to decreased soil fertility and nitrate enrichment of downstream surface and groundwaters.
Denitrification
NO3– N2+ N2O Through denitrification, oxidized forms of nitrogen such as nitrate and nitrite (NO2–) are converted to dinitrogen (N2) and, to a lesser extent, nitrous oxide gas. Denitrification is an anaerobic process that is carried out by denitrifying bacteria, which convert nitrate to dinitrogen in the following sequence:
NO3– NO2– NO N2O N2.
Nitric oxide and nitrous oxide are both environmentally important gases. Nitric oxide (NO) contributes to smog, and nitrous oxide (N2O) is an important greenhouse gas, thereby contributing to global climate change.
Once converted to dinitrogen, nitrogen is unlikely to be reconverted to a biologically available form because it is a gas and is rapidly lost to the atmosphere. Denitrification is the only nitrogen transformation that removes nitrogen from ecosystems (essentially irreversibly), and it roughly balances the amount of nitrogen fixed by the nitrogen fixers described above.
Denitrification of Nitrate-N
Certain soil bacteria that thrive in saturated (anaerobic) soil conditions will convert nitrate-N to oxygen and nitrogen gases. Volatilization of the nitrogen gas can result in N losses of as much as 5% of the available nitrate-N per day. Soils at greatest risk to denitrification N loss are those that are naturally heavy and poorly drained, plus fields with significant levels of soil compaction that restricts natural drainage. Because denitrification affects nitrate-N, the relative risk of N fertilizer products is identical to that for leaching N loss (Fig.16.).
Nitrogen immobilization
A fourth N loss mechanism is more temporary in nature. Soil microbes that decompose high carbon-content plant residues to organic matter use soil N during the decomposition process. Consequently, the nitrogen from the surface-applied fertilizer is “tied up” in the resulting organic matter and is temporarily unavailable for plant uptake until mineralization of the organic matter occurs at a later date. Such immobilization of soil N can be especially prevalent in high-residue no-till cropping systems. Unfortunately, applying N fertilizer in the fall to corn residues has not been shown to reduce N immobilization or speed residue decomposition. (http://www.agry.purdue.edu/ext/pubs/2006NLossMechanisms.pdf)

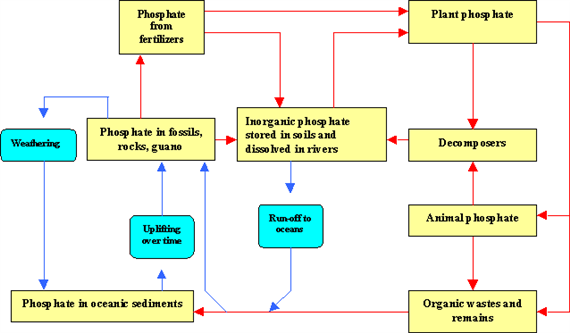
Phosphorus cycle
Phosphorus is an essential nutrient for plants and animals in the form of ions PO43- and HPO42-. It is a part of DNA-molecules, of molecules that store energy (ATP and ADP) and of fats of cell membranes. Phosphorus is also a building block of certain parts of the human and animal body, such as the bones and teeth.
Phosphorus can be found on earth in water, soil and sediments. Unlike the compounds of other matter cycles phosphorus cannot be found in air in the gaseous state. This is because phosphorus is usually liquid at normal temperatures and pressures. It is mainly cycling through water, soil and sediments. In the atmosphere phosphorus can mainly be found as very small dust particles.
Phosphorus moves slowly from deposits on land and in sediments, to living organisms, and than much more slowly back into the soil and water sediment. The phosphorus cycle is the slowest one of the matter cycles that are described here.
Phosphorus is most commonly found in rock formations and ocean sediments as phosphate salts. Phosphate salts that are released from rocks through weathering usually dissolve in soil water and will be absorbed by plants. Because the quantities of phosphorus in soil are generally small, it is often the limiting factor for plant growth. That is why humans often apply phosphate fertilizers on farmland. Phosphates are also limiting factors for plant-growth in marine ecosystems, because they are not very water-soluble. Animals absorb phosphates by eating plants or plant-eating animals.
Phosphorus cycles through plants and animals much faster than it does through rocks and sediments. When animals and plants die, phosphates will return to the soils or oceans again during decay. After that, phosphorus will end up in sediments or rock formations again, remaining there for millions of years. Eventually, phosphorus is released again through weathering and the cycle starts over (Fig. 17).
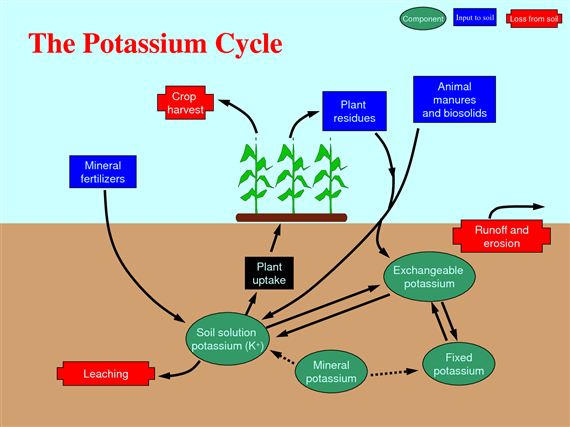
Potassium cycle
Potassium is taken up by plants in large quantities and is necessary to many plant functions, including carbohydrates metabolism, enzyme activation, osmotic regulation, and protein synthesis. Potassium is essential for photosynthesis, for nitrogen fixation in legumes, starch formation, and translocation of sugars. As a result of several of these functions, a good supply of potassium promotes production of plump grains and large tubers.
Potassium is important in helping plants adapt to environmental stresses (e.g. improved drought tolerance and winter hardiness, better resistance to fungal diseases and insect pests (Fig. 64).
Eutrophication
It means the gradual increase in the concentration of phosphorus, nitrogen, and other plant nutrients in an aging aquatic ecosystem such as a lake. The productivity or fertility of such an ecosystem increases as the amount of organic material that can be broken down into nutrients increases. This material enters the ecosystem primarily by runoff from land that carries debris and products of the reproduction and death of terrestrial organisms. Blooms, or great concentrations of algae and microscopic organisms, often develop on the surface, preventing the light penetration and oxygen absorption necessary for underwater life.
Soil science
Prof. Blaskó Lajos (2008), Soil Science
==============================================================
Figure 2.10 Cation uptake can have an acidic effect

Figure 2.12 Anion uptake can have a basic effect
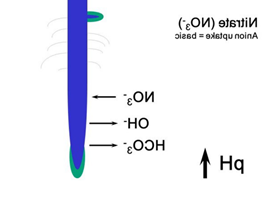
Figure 2.11 Co-absorption of cations and anions at the same rate does not lead to a change in pH.

UF-AG Extension
—————————————————————
INTRODUCTION Iron is an essential nutrient for plants and serves as a cofactor for a wide variety of cellular processes, such as oxygen transport, cellular respiration, chlorophyll biosynthesis, thylakoid biogenesis and chloroplast development (Kobayashi and Nishizawa, 2012; Tanuja Poonia et al, 2018). The availability of Fe is severely limited in calcareous soils due to their low solubility at high pH and bicarbonate concentration which reduces the Fe uptake by inactivating the Fe in plants (Mortvedt, 1991; Najafi-Ghiri et al., 2013). Hence, Fe-deficiency induced chlorosis is a serious problem resulted in the yield loss and quality of crop produces in many crops particularly in the calcareous soils (Kim and Guerinot, 2007; Zheng, 2010). It is also closely related to the prevalence of Fe-deficiency-induced anemia in human beings (Murgia et al., 2012). Amelioration of Fe deficiency in soils and plants was generally achieved through the use of inorganic Fe salts, Fe chelates, organic manures, etc. either through soil application or as a foliar spray which differs significantly in maintaining soil Fe availability. Addition of Fe chelates to calcareous soils was proved to be very effective in maintaining soil solution Fe and the efficacy was better with Fe- EDDHA, Fe EDTA and Fe-DTPA but the recovery of Fe from ferrous sulphate was negligible under high soil pH and calcareousness (Jaloud et al., 2013; Faraz et al., 2014; Sedigheh Safarzadeh et al., 2018). Inclusion of organic manures proved to be beneficial in increasing the availability of Fe in soils and it was widely reported by many researchers (Ali et al., 2007; Yunchen Zhao, 2009; Amin, 2018). Further the Fe availability significantly correlated with many soil properties particularly pH, carbonate and bicarbonate ions and organic carbon content which majorly controls the availability (Obrador et al., 2007; Wang et al., 2009; Wu et al., 2010; Canasveras et al., 2014; Mahendra Kumar et al., 2017). Hence the present study was taken up to test the effectiveness of various levels and sources of Fe on Fe availability in calcareous and noncalcareous soils with and without amendments.
…
CONCLUSION To conclude, a linear increase in DTPA Fe extractability was observed with incubation period in all three soils and the highest availability was associated with the addition of 10 kg Fe EDTA ha-1 followed by 50 kg FeSO4 ha-1. Inclusion of FYM at 12.5 t ha-1 and 0.25 % Acetic acid considerably improved the Fe availability in soils and the better effect was registered with FYM. Higher Fe extractability was noted up to 30 days in the red calcareous soil while in black calcareous soils the release was linear up to 45 days. A negative correlation between soil pH and calcareousness was observed on the Fe availability in the soil
Calcareous soils (containing free lime) are common in many arid and semi-arid re gions af North America and occur as inclusions in mare humid regions. Phosphorus
(P) is very reactive with lime. Fallowing fertilizer application, P undergoes a series af reactions that gradually reduce its solubility. In mast calcareous sails, there does not appear ta be a strong agronomic advantage af any particular P source when managed properly. Organic matter can inhibit P fixation reactions ta same extent. SOILS fertilizer recommendations call far additional P ta be added when the sail contains high amounts af free lime. a lc ar eous soils are common in arid and semi-arid climates and occur as inclusions in more humid regions, af fecting over 1.5 billion acres of soil world wide and comprising more than 17% of the soils in the U.S. Calcareous soils are identi fied by the presence of the mineral calcium carbonate (CaC0 3 or lime) in the parent material and an accumulation of lime. This is most easily recognized by the efferves
cence (fizzing) that occurs when these soils are treated with dilute acid. The pH of these soils is usually above 7 and may be as high as 8.5. When these soils contain sodium carbonate, the pH may exceed 9. In some soils, CaC0 3 can concentrate into very hard layers, termed caliche, that are impermeable to water and plant roots. Calcareous soils can be extremely pro
surfaces, and pre cipit a ti onof various calcium phosphate min erals. While the total lime con tent of a soil is important for predicting P re actions, the lime particle size (and its effect on reac tive surface area) is often a better predictor of P behavior. Al though a calcar eous soil may be dominated by Effervescence (fizzing) occurs when colcoreous soils ore treoted with dilute ocid. Regulor soil testing is importont to monitor ovoilobility of P in colcoreous soils. ductive for agricultural use when they are managed properly. Since they are most fre quently found in semi-arid and arid re gions, supplemental irrigation water is of ten the frrst barrier for crop production. Limited availability of Pis often the next most limiting factor for plant growth. When P fertilizer is added to calcare ous soils, a series of fixation reactions oc cur that gradually decrease its solubility and eventually its availability to plants. Phosphorus “fixation” is a combination of surface adsorption on both clay and lime free lime, it may also contain significant amounts of iron (Fe), aluminum (Al), and manganese (Mn)… either as discrete min erals, as coatings on soil particles, or complexed with soil organic matter. These metals provide strong sorption sites for P and are frequently more significant in con trolling P solubility in calcareous soils than lime itself. Their importance should not be ignored. As fertilizer P reacts in calcareous soils, it is converted to less soluble compounds such as dicalcium phosphate dihydrate or

Figure 1. Fertilizer P undergoes a reduction in solubility following addition to three calcareous soils (Sharpley et al., 1989).
octacalcium phosphate. In some cases it may eventually convert to hydroxyapatite. A variety of management practices can he used to slow these natural fixation pro cesses and increase the efficiency of applied fertilizer for crop growth. A number of the factors controlling P availability will he briefly covered. Time—ln soluhle rock Pis treated af ter mining from geologic deposits to en hance its solubility and usefulness for plants. Fertilizer P is most soluble imme diately after addition to soil, then it un dergoes many chemical reactions that re sult in gradually diminished solubility (Figure 1).
Residual fertilizer P continues to he available for plant uptake for many years, but freshly applied P is generally most soluble and available for plant uptake. The common practice of building soil P concentrations to appropriate agronomic ranges provides a long-term source of this nutrient to crops. Phosphorus Fertilizer Source—Many studies have demonstrated that there are no consistent agronomic differences in most commercially available P fertilizers added to calcareous soils. The selection of a specific P source should he based on other factors such as application equipment, suitability of fluids or granules, and price. However, considerable work is cur rently underway to improve P availability with new P products and fertilizer addi tives. This topic will he explored in greater detail in future articles. For example, re cent work from Australia in extremely cal careous soils has suggested that fluid P sources may have somewhat greater solu bility and enhanced plant availability than granular fertilizers. It has been hypoth esized that granule dissolution may he sup pressed in these soil conditions. Additional work is underway in the U.S. to see if these results hold for soil conditions more typi cal of North America There is large variability in the solu bility and availability of P from various materials added to calcareous soil (Figure 2). These large differences are largely due to the unique properties of the materials, rather than any unique character associ ated with a specific soil. For example, the polymer-coated, slow release P source has very low apparent solubility, but is able to sup port high levels of plant P ac cumula tio n. The soluble P sources and liquid manures have very high solubility and also are able to maintain high P recovery by barley. Organic Matt e1′- l n the soil solution, there are several chemical components that will delay or prevent the reaction of P with lime. Organic mat ter has been found to interfere in the fixation reactions of P with lime. This inhibition of P
Figure 2. Extractability and P uptake by barley from various sources following incubation in a 12% lime soil. Sources initially added at a rate of 60 mg P/kg; extractions are average of 2 and 6-weeks sampling dates. (Leytem and Westerman, 2005).
Figure 3. The effect of soil temperature on fertilizer P extractability in a calcareous soil µavid and Rowell, 2003).
fixation may account for the observation that P availability is frequently greater in manured soils and with the addition of humic substances in lime-rich soil. Higher levels of soluble Fe, Al, and Mn are also related to increased P fixation in calcare ous soils. Temperature Soil temperature has two opposing effects on soil P availability. When fertilizer P is added to soil, it con tinually reacts and forms increasingly stable compounds for many months after application. The kinetics of the conversion of P to less soluble forms is more rapid under warmer conditions than incooler soil (Figure 3). An opposite effect occurs as increased soil temperature raises the solubility of soil P forms (both adsorbed or precipitated P). This well-known phenomenon accounts for frequent crop responses from added P in cool soils in the spring. In addition to im proved solubility, higher soil temperature increases P diffusion to plant roots and en hances overall root activity and prolifera tion. When planting early in the season, or in high-residue conditions, cold soil tem peratures can induce an early-stage P de ficiency in many types of soil. A starter P fertilizer application may help overcome these limitations. Adj11sting for Calcareous Soil s—Since the presence of lime in soils can reduce P availability to crops, fertilizer recommen dations are frequently adjusted to account for this condition. For example, the Uni versity of Idaho recommendations for potat rtilization state that an additional 10 lb Pp/A needs to he applied for every 1% increase in soil lime (Figure 4).
Figure 4. University of Idaho P fertilizer recom mendations for potatoes grown in calcareous soil take into account the free lime content of the soil (Tindall and Stark, 1997).
Calcareous soils can he extremely productive when managed properly. Maintaining an adequate supply of plant available P is essential to profitable and sustainable crop production. Since a vari ety of soil reactions tend to decrease the plant-availability of added fertilizer P in calcareous soil, regular soil testing should he conducted to avoid crop loss due to plant nutrient deficiency. ‘!ffl
The Nature of Phosphorus in Calcareous Soils, By A.B. Leytem and R.L. Mikkelsen, pdf
