Clay and CEC
Montmorillonite
| Molecular Formula | Al2H2O12Si4 |
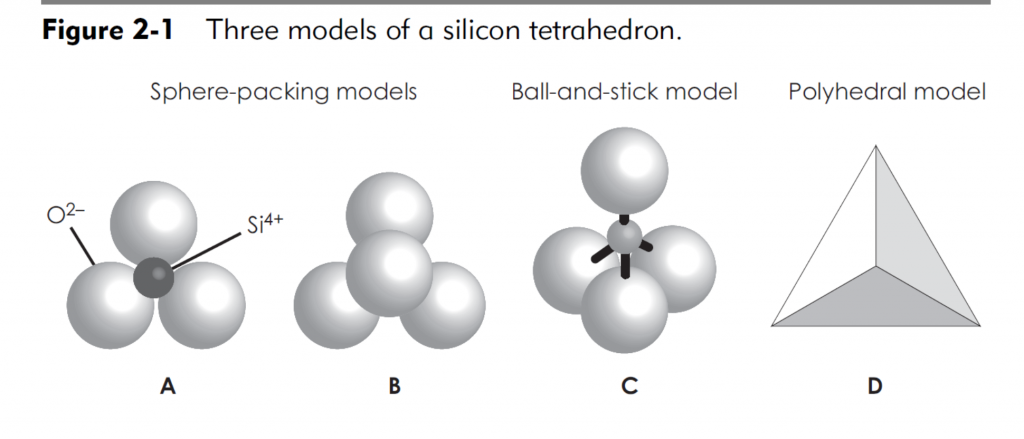
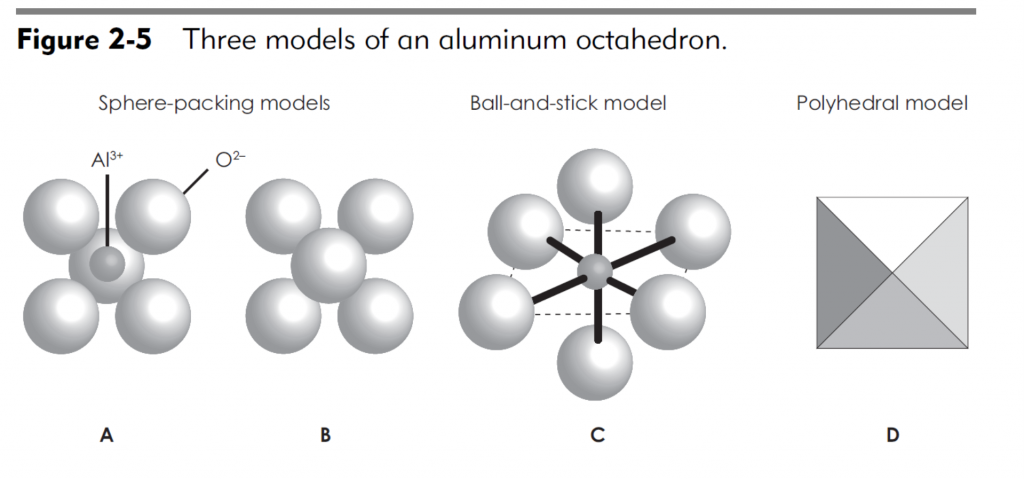

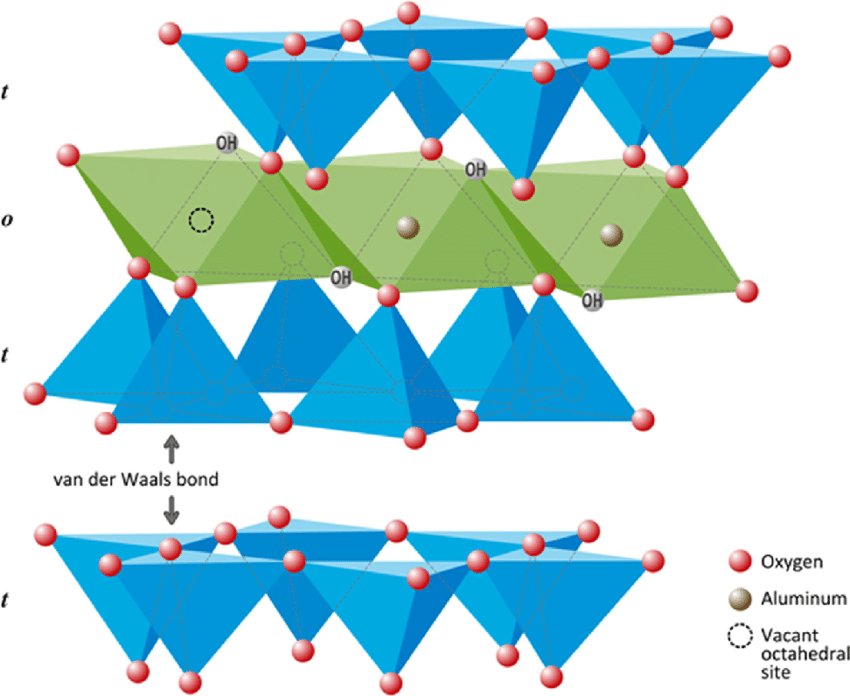
The basic structural unit of montmorillonite mineral consists of one alumina sheet sandwiched between two silica sheets. The oxygen atoms at the tip of each silica sheet combine with the OH ions of both sides of the alumina sheet by hydrogen bond. As the bond is fairly strong, the basic structural unit of montmorillonite mineral is also stable. The thickness of the basic structural unit of montmorillonite is 9.2 Å.
Montmorillonite mineral is formed by stacking of several structural units one over the other. The interface between the two structural units during this stacking is through the silica sheet of one structural unit and the silica sheet of the other unit.
This bond is by van der Waals’ forces, which is rather weak. In dry condition, the van der Waals’ forces are rather strong so that it is difficult to break a dry lump of montmorillonite clay. However, water can easily enter between the layers, often dividing the montmorillonite particle into individual structural units.
One characteristic of van der Waals’ forces is that their magnitude decreases rapidly with distance. As water enters between the structural units, the distance between them increases, causing reduction in attractive forces. Thus, a montmorillonite clay particle is stable as long as it is dry but breaks into individual structural units in the presence of water.
————
Cation Exchange:


In a near neutral soil, calcium remains adsorbed on colloidal particle. Hydrogen ion (H+ ) generated as organic and mineral acids formed due to decomposition of organic matter. In colloid, hydrogen ion is adsorbed more strongly than is the calcium (Ca++). The reaction takes place rapidly and the interchange of calcium and hydrogen is Chemically equivalent.
This phenomenon of the exchange of cations between soil and salt solution is known as Cation exchange or Base exchange and the cations that take part in this reaction are called exchangeable cations. Cation exchange reactions are reversible.
Hence, if some form of limestone or other basic calcium compound is applied to an acid soil, the reverse of the replacement just given above occurs. The active calcium ions replace the hydrogen and other cations by mass action. As a result, the clay becomes higher in exchangeable calcium and lower in adsorbed hydrogen and aluminium.
If a soil is treated with a liberal application of a fertilizer containing potassium chloride, following reaction may occur: ; then:
Some of the added potassium pushes its way into the colloidal complex and forces out equivalent quantities of calcium, hydrogen and other elements (e.g., M) which appear in the soil solution. The adsorption of the added potassiumis largely in an available condition. Hence, cation exchange is an important consideration for making already present nutrients in soils available to plants. Cation exchange also makes available the nutrients, applied in commercial fertilizers.
Cation Exchange Capacity (C.E.C.):
The cation exchange capacity of a soil represents the capacity of the colloidal complex to exchange all its cations with the cations of the electrolyte solution (surrounding liquid). It also represents the total cation adsorbing capacity of a soil. Cation exchange in most soils increases with pH. At a very low pH value, C.E.C. is higher and at high pH, C.E.C. is relatively lower.
————-
SOIL, ROOTS, AND MICROBES
The soil is a complex physical, chemical, and biological substrate. It is a heterogeneous material containing solid, liquid, and gaseous phases (see Chapter 4). All of these phases interact with mineral elements. The inorganic particles of the solid phase provide a reservoir of potassium, calcium, magnesium, and iron. Also associated with this solid phase are organic compounds containing nitrogen, phosphorus, and sulfur, among other elements. The liquid phase of the soil constitutes the soil solution, which contains dissolved mineral ions and serves as the medium for ion movement to the root surface. Gases such as oxygen, carbon dioxide, and nitrogen are dissolved in the soil solution, but in roots gases are exchanged predominantly through the air gaps between soil particles. From a biological perspective, soil constitutes a diverse ecosystem in which plant roots and microorganisms compete strongly for mineral nutrients. In spite of this competition, roots and microorganisms can form alliances for their mutual benefit (symbioses, singular symbiosis). In this section we will discuss the importance of soil properties, root structure, and mycorrhizal symbiotic relationships to plant mineral nutrition. Chapter 12 addresses symbiotic relationships with nitrogen-fixing bacteria.
Negatively Charged Soil Particles Affect the Adsorption of Mineral Nutrients
Soil particles, both inorganic and organic, have predominantly negative charges on their surfaces. Many inorganic soil particles are crystal lattices that are tetrahedral arrangements of the cationic forms of aluminum and silicon (Al3+ and Si4+) bound to oxygen atoms, thus forming aluminates and silicates. When cations of lesser charge replace Al3+ and Si4+, inorganic soil particles become negatively charged. Organic soil particles originate from the products of the microbial decomposition of dead plants, animals, and microorganisms. The negative surface charges of organic particles result from the dissociation of hydrogen ions from the carboxylic acid and phenolic groups present in this component of the soil. Most of the world’s soil particles, however, are inorganic. Inorganic soils are categorized by particle size:
• Gravel has particles larger than 2 mm.
• Coarse sand has particles between 0.2 and 2 mm.
• Fine sand has particles between 0.02 and 0.2 mm.
• Silt has particles between 0.002 and 0.02 mm.
• Clay has particles smaller than 0.002 mm (see Table 4.1).
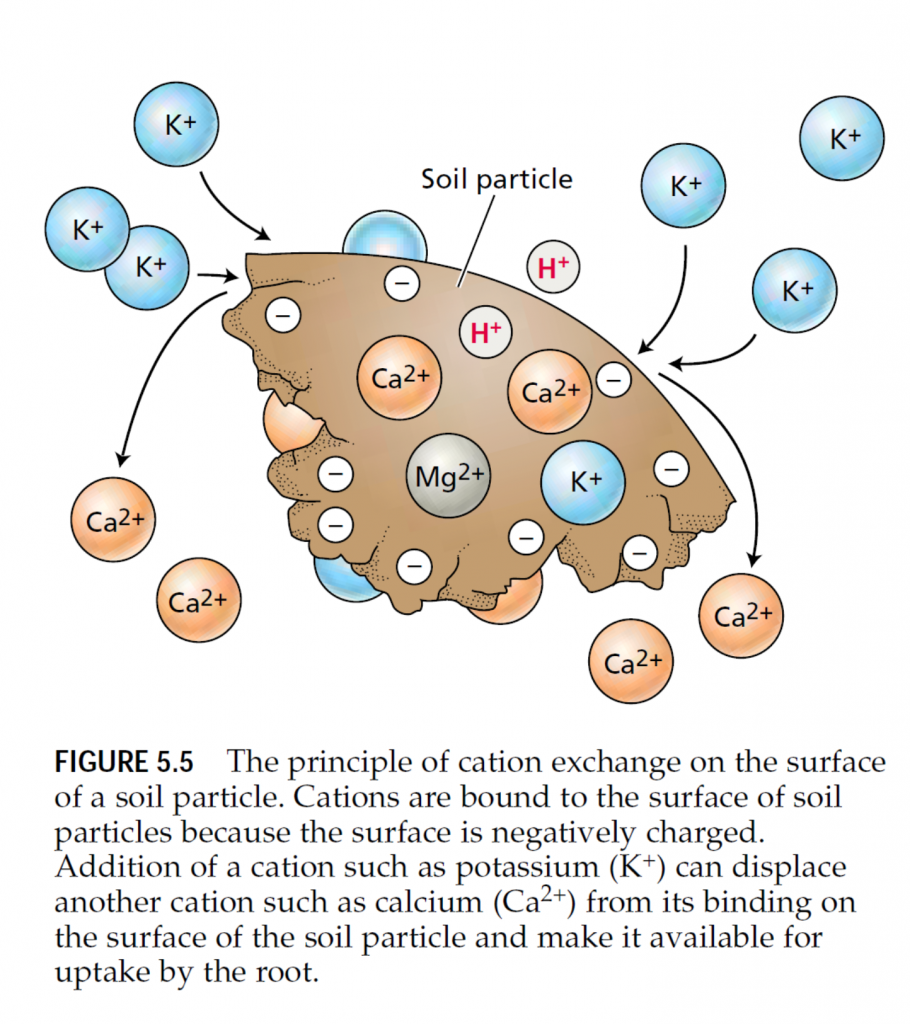
The silicate-containing clay materials are further divided into three major groups—kaolinite, illite, and montmorillonite— based on differences in their structure and physical properties (Table 5.5). The kaolinite group is generally found in well-weathered soils; the montmorillonite and illite groups are found in less weathered soils. Mineral cations such as ammonium (NH4 +) and potassium (K+) adsorb to the negative surface charges of inorganic and organic soil particles. This cation adsorption is an important factor in soil fertility. Mineral cations adsorbed on the surface of soil particles are not easily lost when the soil is leached by water, and they provide a nutrient reserve available to plant roots. Mineral nutrients adsorbed in this way can be replaced by other cations in a process known as cation exchange (Figure 5.5). The degree to which a soil can adsorb and exchange ions is termed its cation exchange capacity (CEC) and is highly dependent on the soil type. A soil with higher cation exchange capacity generally has a larger reserve of mineral nutrients. Mineral anions such as nitrate (NO3 –) and chloride (Cl–) tend to be repelled by the negative charge on the surface of soil particles and remain dissolved in the soil solution. Thus the anion exchange capacity of most agricultural soils is small compared to the cation exchange capacity. Among anions, nitrate remains mobile in the soil solution, where it is susceptible to leaching by water moving through the soil. Phosphate ions (H2PO2 –) may bind to soil particles containing aluminum or iron because the positively charged iron and aluminum ions (Fe2+, Fe3+, and Al3+) have hydroxyl (OH–) groups that exchange with phosphate. As a result, phosphate can be tightly bound, and its mobility and availability in soil can limit plant growth. Sulfate (SO4 2–) in the presence of calcium (Ca2+) forms gypsum (CaSO4). Gypsum is only slightly soluble, but it releases sufficient sulfate to support plant growth.
Most nonacid soils contain substantial amounts of calcium; consequently, sulfate mobility in these soils is low, so sulfate is not highly susceptible to leaching.

Soil pH Affects Nutrient Availability, Soil Microbes, and Root Growth
Hydrogen ion concentration (pH) is an important property of soils because it affects the growth of plant roots and soil microorganisms. Root growth is generally favored in slightly acidic soils, at pH values between 5.5 and 6.5. Fungi generally predominate in acidic soils; bacteria become more prevalent in alkaline soils. Soil pH determines the availability of soil nutrients (see Figure 5.4). Acidity promotes the weathering of rocks that releases K+, Mg2+, Ca2+, and Mn2+ and increases the solubility of carbonates, sulfates, and phosphates. Increasing the solubility of nutrients facilitates their availability to roots. Major factors that lower the soil pH are the decomposition of organic matter and the amount of rainfall. Carbon dioxide is produced as a result of the decomposition of organic material and equilibrates with soil water in the following reaction: CO2 + H2O ~ H+ + HCO3 – This reaction releases hydrogen ions (H+), lowering the pH of the soil. Microbial decomposition of organic material also produces ammonia and hydrogen sulfide that can be oxidized in the soil to form the strong acids nitric acid (HNO3) and sulfuric acid (H2SO4), respectively. Hydrogen ions also displace K+, Mg2+, Ca2+, and Mn2+ from the cation exchange complex in a soil. Leaching then may remove these ions from the upper soil layers, leaving a more acid soil. By contrast, the weathering of rock in arid regions releases K+, Mg2+, Ca2+, and Mn2+ to the soil, but because of the low rainfall, these ions do not leach from the upper soil layers, and the soil remains alkaline.

Excess Minerals in the Soil Limit Plant Growth
When excess minerals are present in the soil, the soil is said to be saline, and plant growth may be restricted if these mineral ions reach levels that limit water availability or exceed the adequate zone for a particular nutrient (see Chapter 25). Sodium chloride and sodium sulfate are the most common salts in saline soils. Excess minerals in soils can be a major problem in arid and semiarid regions because rainfall is insufficient to leach the mineral ions from the soil layers near the surface. Irrigated agriculture fosters soil salinization if insufficient water is applied to leach the salt below the rooting zone. Irrigation water can contain 100 to 1000 g of minerals per cubic meter. An average crop requires about 4000 m3 of water per acre. Consequently, 400 to 4000 kg of minerals may be added to the soil per crop (Marschner 1995). In saline soil, plants encounter salt stress. Whereas many plants are affected adversely by the presence of relatively low levels of salt, other plants can survive high levels (salt-tolerant plants) or even thrive (halophytes) under such conditions. The mechanisms by which plants tolerate salinity are complex (see Chapter 25), involving molecular synthesis, enzyme induction, and membrane transport. In some species, excess minerals are not taken up; in others, minerals are taken up but excreted from the plant by salt glands associated with the leaves. To prevent toxic buildup of mineral ions in the cytosol, many plants may sequester them in the vacuole (Stewart and Ahmad 1983). Efforts are under way to bestow salt tolerance on salt-sensitive crop species using both classic plant breeding and molecular biology (Hasegawa et al. 2000). Another important problem with excess minerals is the accumulation of heavy metals in the soil, which can cause severe toxicity in plants as well as humans . Heavy metals include zinc, copper, cobalt, nickel, mercury, lead, cadmium, silver, and chromium (Berry and Wallace 1981).
Plants Develop Extensive Root Systems
The ability of plants to obtain both water and mineral nutrients from the soil is related to their capacity to develop an extensive root system. In the late 1930s, H. J. Dittmer examined the root system of a single winter rye plant after 16 weeks of growth and estimated that the plant had 13 × 106 primary and lateral root axes, extending more than 500 km in length and providing 200 m2 of surface area (Dittmer 1937). This plant also had more than 1010 root hairs, providing another 300 m2 of surface area the desert, the roots of mesquite (genus Prosopis) may extend down more than 50 m to reach groundwater. Annual crop plants have roots that usually grow between 0.1 and 2.0 m in depth and extend laterally to distances of 0.3 to 1.0 m. In orchards, the major root systems of trees planted 1 m apart reach a total length of 12 to 18 km per tree. The annual production of roots in natural ecosystems may easily surpass that of shoots, so in many respects, the aboveground portions of a plant represent only “the tip of an iceberg.” Plant roots may grow continuously throughout the year. Their proliferation, however, depends on the availability of water and minerals in the immediate microenvironment surrounding the root, the so-called rhizosphere. If the rhizosphere is poor in nutrients or too dry, root growth is slow. As rhizosphere conditions improve, root growth increases. If fertilization and irrigation provide abundant nutrients and water, root growth may not keep pace with shoot growth. Plant growth under such conditions becomes carbohydrate limited, and a relatively small root system meets the nutrient needs of the whole plant (Bloom et al. 1993). Roots growing below the soil surface are studied by special techniques

Root Systems Differ in Form but Are Based on Common Structures
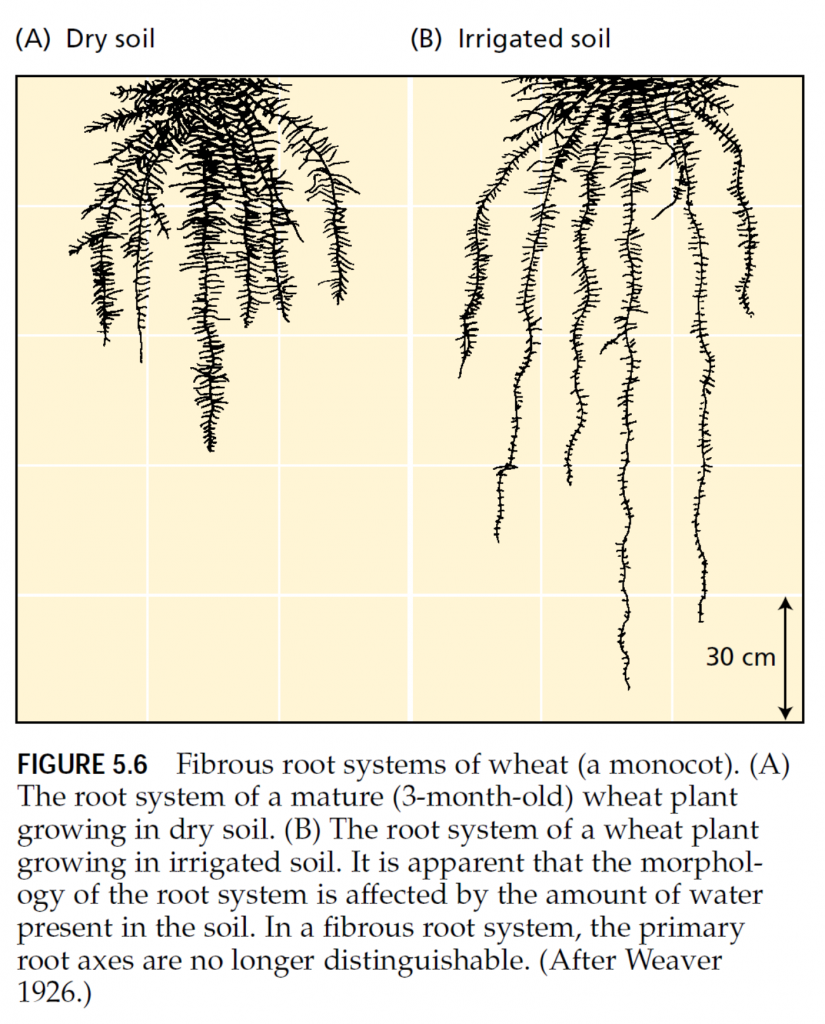
The form of the root system differs greatly among plant species. In monocots, root development starts with the emergence of three to six primary (or seminal) root axes from the germinating seed. With further growth, the plant extends new adventitious roots, called nodal roots or brace roots. Over time, the primary and nodal root axes grow and branch extensively to form a complex fibrous root system (Figure 5.6). In fibrous root systems, all the roots generally have the same diameter (except where environmental conditions or pathogenic interactions modify the root structure), so it is difficult to distinguish a main root axis. In contrast to monocots, dicots develop root systems with a main single root axis, called a taproot, which may thicken as a result of secondary cambial activity. From this main root axis, lateral roots develop to form an extensively branched root system (Figure 5.7). The development of the root system in both monocots and dicots depends on the activity of the root apical meristem and the production of lateral root meristems. Figure 5.8 shows a generalized diagram of the apical region of a plant root and identifies the three zones of activity: meristematic, elongation, and maturation. In the meristematic zone, cells divide both in the direction of the root base to form cells that will differentiate into the tissues of the functional root and in the direction of the root apex to form the root cap. The root cap protects the delicate meristematic cells as the root moves through the soil. It also secretes a gelatinous material called mucigel, which commonly surrounds the root tip. The precise function of the mucigel is uncertain, but it has been suggested that it lubricates the penetration of the root through the soil, protects the root apex from desiccation, promotes the transfer of nutrients to the root, or affects the interaction between roots and soil microorganisms (Russell 1977). The root cap is central to the perception of gravity, the signal that directs the growth of roots downward. This process is termed the gravitropic response (see Chapter 19). Cell division at the root apex proper is relatively slow; thus this region is called the quiescent center. After a few generations of slow cell divisions, root cells displaced from the apex by about 0.1 mm begin to divide more rapidly. Cell division again tapers off at about 0.4 mm from the apex, and the cells expand equally in all directions. The elongation zone begins 0.7 to 1.5 mm from the apex (see Figure 5.8). In this zone, cells elongate rapidly and undergo a final round of divisions to produce a central ring of cells called the endodermis. The walls of this endodermal cell layer become thickened, and suberin (see Chapter 13) deposited on the radial walls forms the Casparian strip, a hydrophobic structure that prevents the apoplastic movement of water or solutes across the root (see Figure 4.3). The endodermis divides the root into two regions: the cortex toward the outside and the stele toward the inside. The stele contains the vascular elements of the root: the phloem, which transports metabolites from the shoot to the root, and the xylem, which transports water and solutes to the shoot.
Phloem develops more rapidly than xylem, attesting to the fact that phloem function is critical near the root apex. Large quantities of carbohydrates must flow through the phloem to the growing apical zones in order to support cell division and elongation. Carbohydrates provide rapidly growing cells with an energy source and with the carbon skeletons required to synthesize organic compounds. Sixcarbon sugars (hexoses) also function as osmotically active solutes in the root tissue. At the root apex, where the phloem is not yet developed, carbohydrate movement depends on symplastic diffusion and is relatively slow (Bret-Harte and Silk 1994). The low rates of cell division in the quiescent center may result from the fact that insufficient carbohydrates reach this centrally located region or that this area is kept in an oxidized state Root hairs, with their large surface area for absorption of water and solutes, first appear in the maturation zone (see Figure 5.8), and it is here that the xylem develops the capacity to translocate substantial quantities of water and solutes to the shoot.
Different Areas of the Root Absorb Different Mineral Ions
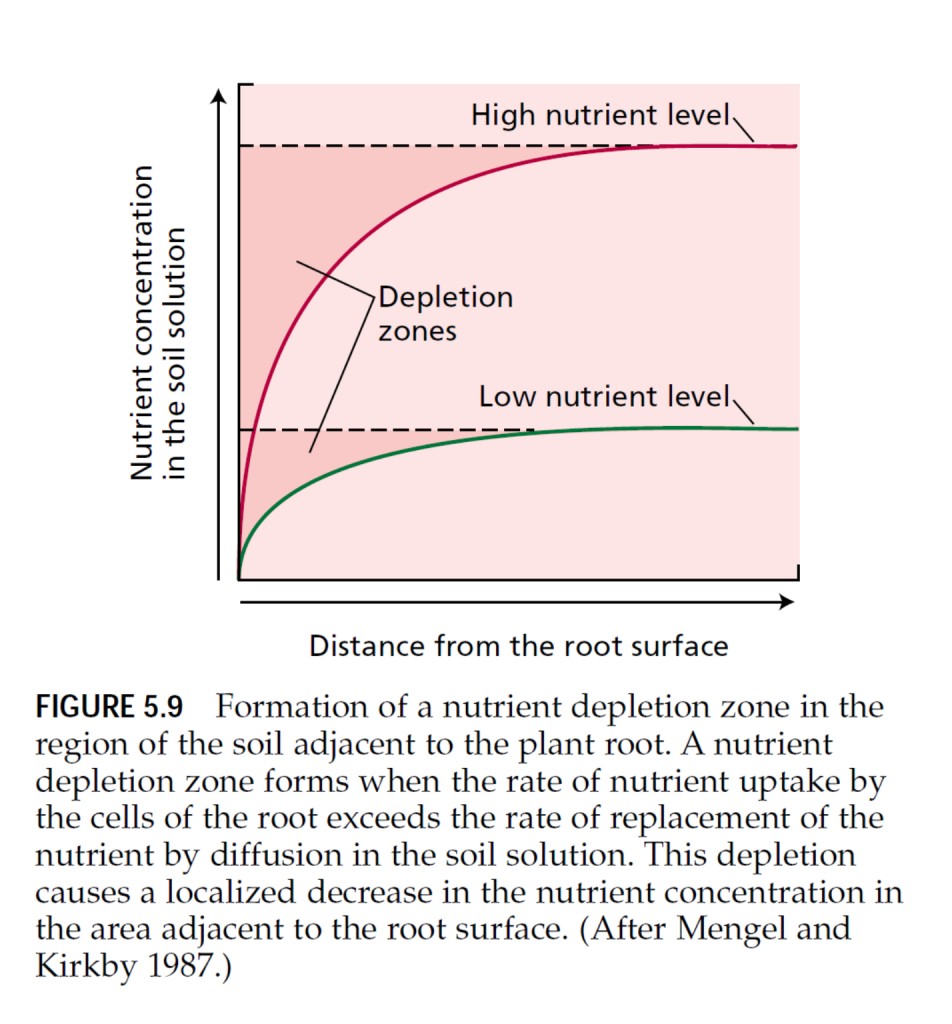
The precise point of entry of minerals into the root system has been a topic of considerable interest. Some researchers have claimed that nutrients are absorbed only at the apical regions of the root axes or branches (Bar-Yosef et al. 1972); others claim that nutrients are absorbed over the entire root surface (Nye and Tinker 1977). Experimental evidence supports both possibilities, depending on the plant species and the nutrient being investigated: • Root absorption of calcium in barley appears to be restricted to the apical region. • Iron may be taken up either at the apical region, as in barley (Clarkson 1985), or over the entire root surface, as in corn (Kashirad et al. 1973). • Potassium, nitrate, ammonium, and phosphate can be absorbed freely at all locations of the root surface (Clarkson 1985), but in corn the elongation zone has the maximum rates of potassium accumulation (Sharp et al. 1990) and nitrate absorption (Taylor and Bloom 1998). • In corn and rice, the root apex absorbs ammonium more rapidly than the elongation zone does (Colmer and Bloom 1998). • In several species, root hairs are the most active in phosphate absorption (Fohse et al. 1991). The high rates of nutrient absorption in the apical root zones result from the strong demand for nutrients in these tissues and the relatively high nutrient availability in the soil surrounding them. For example, cell elongation depends on the accumulation of solutes such as potassium, chloride, and nitrate to increase the osmotic pressure within the cell (see Chapter 15). Ammonium is the preferred nitrogen source to support cell division in the meristem because meristematic tissues are often carbohydrate limited, and the assimilation of ammonium consumes less energy than that of nitrate (see Chapter 12). The root apex and root hairs grow into fresh soil, where nutrients have not yet been depleted. Within the soil, nutrients can move to the root surface both by bulk flow and by diffusion (see Chapter 3). In bulk flow, nutrients are carried by water moving through the soil toward the root. The amount of nutrient provided to the root by bulk flow depends on the rate of water flow through the soil toward the plant, which depends on transpiration rates and on nutrient levels in the soil solution. When both the rate of water flow and the concentrations of nutrients in the soil solution are high, bulk flow can play an important role in nutrient supply. In diffusion, mineral nutrients move from a region of higher concentration to a region of lower concentration. Nutrient uptake by the roots lowers the concentration of nutrients at the root surface, generating concentration gradients in the soil solution surrounding the root. Diffusion of nutrients down their concentration gradient and bulk flow resulting from transpiration can increase nutrient availability at the root surface. When absorption of nutrients by the roots is high and the nutrient concentration in the soil is low, bulk flow can supply only a small fraction of the total nutrient requirement (Mengel and Kirkby 1987). Under these conditions, diffusion rates limit the movement of nutrients to the root surface. When diffusion is too slow to maintain high nutrient concentrations near the root, a nutrient depletion zone forms adjacent to the root surface (Figure 5.9). This zone extends from about 0.2 to 2.0 mm from the root surface, depending on the mobility of the nutrient in the soil. The formation of a depletion zone tells us something important about mineral nutrition: Because roots deplete the mineral supply in the rhizosphere, their effectiveness in mining minerals from the soil is determined not only by the rate at which they can remove nutrients from the soil solution, but by their continuous growth. Without growth, roots would rapidly deplete the soil adjacent to their surface. Optimal nutrient acquisition therefore depends both on the capacity for nutrient uptake and on the ability of the root system to grow into fresh soil.
Mycorrhizal Fungi Facilitate Nutrient Uptake by Roots
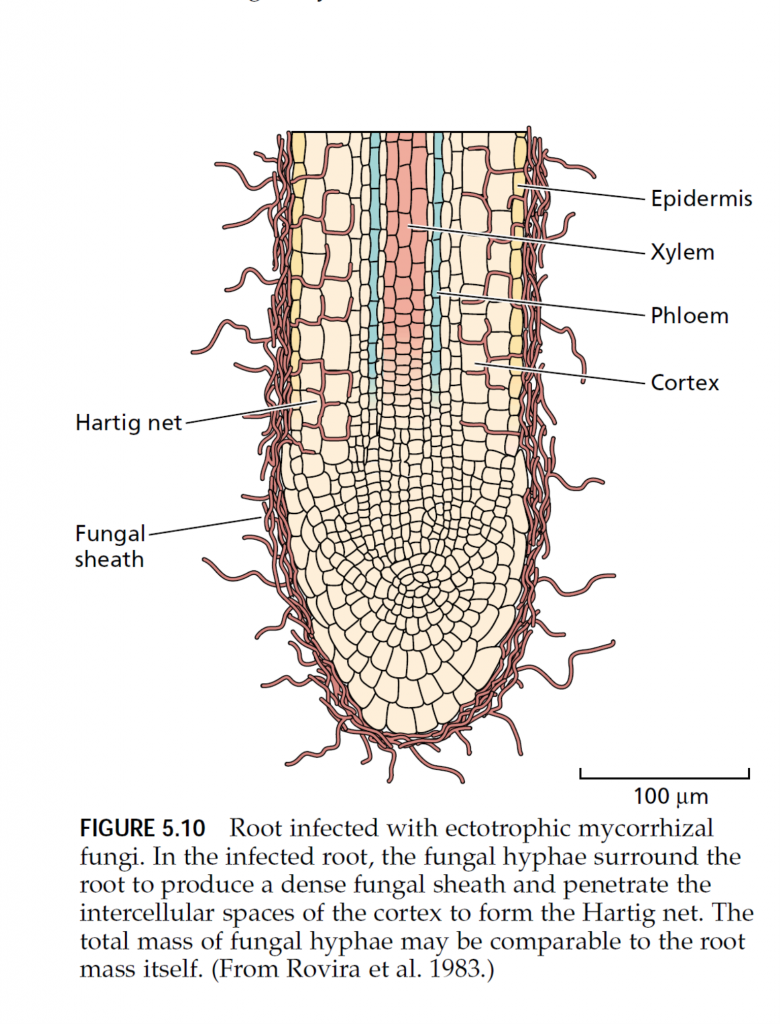
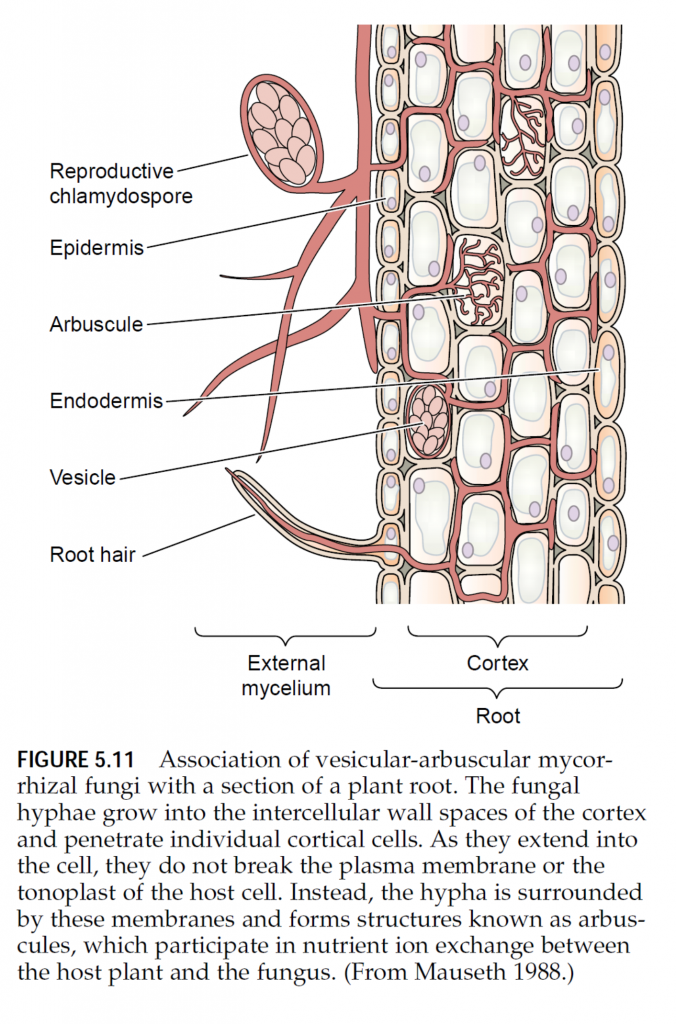
Our discussion thus far has centered on the direct acquisition of mineral elements by the root, but this process may be modified by the association of mycorrhizal fungi with the root system. Mycorrhizae (singular mycorrhiza, from the Greek words for “fungus” and “root”) are not unusual; in fact, they are widespread under natural conditions. Much of the world’s vegetation appears to have roots associated with mycorrhizal fungi: 83% of dicots, 79% of monocots, and all gymnosperms regularly form mycorrhizal associations (Wilcox 1991). On the other hand, plants from the families Cruciferae (cabbage), Chenopodiaceae (spinach), and Proteaceae (macadamia nuts), as well as aquatic plants, rarely if ever have mycorrhizae. Mycorrhizae are absent from roots in very dry, saline, or flooded soils, or where soil fertility is extreme, either high or low. In particular, plants grown under hydroponics and young, rapidly growing crop plants seldom have mycorrhizae. Mycorrhizal fungi are composed of fine, tubular filaments called hyphae (singular hypha). The mass of hyphae that forms the body of the fungus is called the mycelium (plural mycelia). There are two major classes of mycorrhizal fungi: ectotrophic mycorrhizae and vesicular-arbuscular mycorrhizae (Smith et al. 1997). Minor classes of mycorrhizal fungi include the ericaceous and orchidaceous mycorrhizae, which may have limited importance in terms of mineral nutrient uptake. Ectotrophic mycorrhizal fungi typically show a thick sheath, or “mantle,” of fungal mycelium around the roots, and some of the mycelium penetrates between the cortical cells (Figure 5.10). The cortical cells themselves are not penetrated by the fungal hyphae but instead are surrounded by a network of hyphae called the Hartig net. Often the amount of fungal mycelium is so extensive that its total mass is comparable to that of the roots themselves. The fungal mycelium also extends into the soil, away from this compact mantle, where it forms individual hyphae or strands containing fruiting bodies. The capacity of the root system to absorb nutrients is improved by the presence of external fungal hyphae that are much finer than plant roots and can reach beyond the areas of nutrient-depleted soil near the roots (Clarkson 1985). Ectotrophic mycorrhizal fungi infect exclusively tree species, including gymnosperms and woody angiosperms. Unlike the ectotrophic mycorrhizal fungi, vesiculararbuscular mycorrhizal fungi do not produce a compact mantle of fungal mycelium around the root. Instead, the hyphae grow in a less dense arrangement, both within the root itself and extending outward from the root into the surrounding soil (Figure 5.11). After entering the root through either the epidermis or a root hair, the hyphae not only extend through the regions between cells but also penetrate individual cells of the cortex. Within the cells, the hyphae can form oval structures called vesicles and branched structures called arbuscules. The arbuscules appear to be sites of nutrient transfer between the fungus and the host plant. Outside the root, the external mycelium can extend several centimeters away from the root and may contain spore-bearing structures. Unlike the ectotrophic mycorrhizae, vesicular-arbuscular mycorrhizae make up only a small mass of fungal material, which is unlikely to exceed 10% of the root weight. Vesicular-arbuscular mycorrhizae are found in association with the roots of most species of herbaceous angiosperms (Smith et al. 1997). The association of vesicular-arbuscular mycorrhizae with plant roots facilitates the uptake of phosphorus and trace metals such as zinc and copper. By extending beyond the depletion zone for phosphorus around the root, the external mycelium improves phosphorus absorption. Calculations show that a root associated with mycorrhizal fungi can transport phosphate at a rate more than four times higher than that of a root not associated with mycorrhizae (Nye and Tinker 1977). The external mycelium of the ectotrophic mycorrhizae can also absorb phosphate and make it available to the plant. In addition, it has been suggested that ectotrophic mycorrhizae proliferate in the organic litter of the soil and hydrolyze organic phosphorus for transfer to the root (Smith et al. 1997).
Nutrients Move from the Mycorrhizal Fungi to the Root Cells
Little is known about the mechanism by which the mineral nutrients absorbed by mycorrhizal fungi are transferred to the cells of plant roots. With ectotrophic mycorrhizae, inorganic phosphate may simply diffuse from the hyphae in the Hartig net and be absorbed by the root cortical cells. With vesicular-arbuscular mycorrhizae, the situation may be more complex. Nutrients may diffuse from intact arbuscules to root cortical cells. Alternatively, because some root arbuscules are continually degenerating while new ones are forming, degenerating arbuscules may release their internal contents to the host root cells. A key factor in the extent of mycorrhizal association with the plant root is the nutritional status of the host plant. Moderate deficiency of a nutrient such as phosphorus tends to promote infection, whereas plants with abundant nutrients tend to suppress mycorrhizal infection. Mycorrhizal association in well-fertilized soils may shift from a symbiotic relationship to a parasitic one in that the fungus still obtains carbohydrates from the host plant, but the host plant no longer benefits from improved nutrient uptake efficiency. Under such conditions, the host plant may treat mycorrhizal fungi as it does other pathogens (Brundrett 1991; Marschner 1995).
SUMMARY
…
The soil is a complex substrate—physically, chemically, and biologically. The size of soil particles and the cation exchange capacity of the soil determine the extent to which a soil provides a reservoir for water and nutrients. Soil pH also has a large influence on the availability of mineral elements to plants. If mineral elements, especially sodium or heavy metals, are present in excess in the soil, plant growth may be adversely affected. Certain plants are able to tolerate excess mineral elements, and a few species—for example, halophytes in the case of sodium—grow under these extreme conditions. To obtain nutrients from the soil, plants develop extensive root systems. Roots have a relatively simple structure with radial symmetry and few differentiated cell types. Roots continually deplete the nutrients from the immediate soil around them, and such a simple structure may permit rapid growth into fresh soil. Plant roots often form associations with mycorrhizal fungi. The fine hyphae of mycorrhizae extend the reach of roots into the surrounding soil and facilitate the acquisition of mineral elements, particularly those like phosphorus that are relatively immobile in the soil. In return, plants provide carbohydrates to the mycorrhizae. Plants tend to suppress mycorrhizal associations under conditions of high nutrient availability.
